Arginase I-producing myeloid-derived suppressor cells in renal cell carcinoma are a subpopulation of activated granulocytes
- PMID: 19201693
- PMCID: PMC2900845
- DOI: 10.1158/0008-5472.CAN-08-1921
Arginase I-producing myeloid-derived suppressor cells in renal cell carcinoma are a subpopulation of activated granulocytes
Abstract
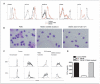
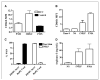
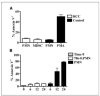
Myeloid-derived suppressor cells (MDSC) producing arginase I are increased in the peripheral blood of patients with renal cell carcinoma (RCC). MDSC inhibit T-cell function by reducing the availability of L-arginine and are therefore considered an important tumor escape mechanism. We aimed to determine the origin of arginase I-producing MDSC in RCC patients and to identify the mechanisms used to deplete extracellular L-arginine. The results show that human MDSC are a subpopulation of activated polymorphonuclear (PMN) cells expressing high levels of CD66b, CD11b, and VEGFR1 and low levels of CD62L and CD16. In contrast to murine MDSC, human MDSC do not deplete L-arginine by increasing its uptake but instead release arginase I into the circulation. Activation of normal PMN induces phenotypic and functional changes similar to MDSC and also promotes the release of arginase I from intracellular granules. Interestingly, although activation of normal PMN usually ends with apoptosis, MDSC showed no increase in apoptosis compared with autologous PMN or PMN obtained from normal controls. High levels of VEGF have been shown to increase suppressor immature myeloid dendritic cells in cancer patients. Treatment of RCC patients with anti-VEGF antibody bevacizumab, however, did not reduce the accumulation of MDSC in peripheral blood. In contrast, the addition of interleukin-2 to the treatment increased the number of MDSC in peripheral blood and the plasma levels of arginase I. These results may provide new insights on the mechanisms of tumor-induced anergy/tolerance and may help explain why some immunotherapies fail to induce an antitumor response.
Figures



References
-
- McDermott DF, Regan MM, Clark JI, et al. Randomized phase III trial of high-dose interleukin-2 versus subcutaneous interleukin-2 and interferon in patients with metastatic renal cell carcinoma. J Clin Oncol. 2005;23:133–41. - PubMed
-
- Schmielau J, Finn OJ. Activated granulocytes and granulocyte-derived hydrogen peroxide are the underlying mechanism of suppression of T-cell function in advanced cancer patients. Cancer Res. 2001;61:4756–60. - PubMed
-
- Zea AH, Rodriguez PC, Atkins MB, et al. Arginase-producing myeloid suppressor cells in renal cell carcinoma patients: a mechanism of tumor evasion. Cancer Res. 2005;65:3044–8. - PubMed
-
- Rodriguez PC, Zea AH, DeSalvo J, et al. l-arginine consumption by macrophages modulates the expression of CD3ζ chain in T lymphocytes. J Immunol. 2003;171:1232–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

