The sequestosome 1/p62 attenuates cytokine gene expression in activated macrophages by inhibiting IFN regulatory factor 8 and TNF receptor-associated factor 6/NF-kappaB activity
- PMID: 19201866
- PMCID: PMC4151355
- DOI: 10.4049/jimmunol.0802755
The sequestosome 1/p62 attenuates cytokine gene expression in activated macrophages by inhibiting IFN regulatory factor 8 and TNF receptor-associated factor 6/NF-kappaB activity
Abstract
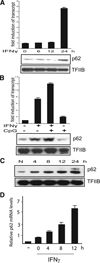
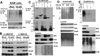
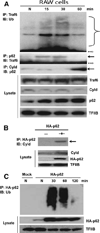
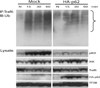
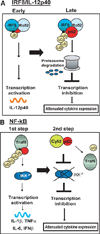
Sequestosome 1/p62 (p62) is a scaffold/adaptor protein with multiple functions implicated for neuronal and bone diseases. It carries a ubiquitin binding domain through which it mediates proteasome-dependent proteolysis. In addition, p62 is reported to regulate NF-kappaB activity in some cells. To date, however, the role of p62 in innate immunity has not been fully elucidated. In this study, we report that IFN-gamma plus TLR signaling stimulates late expression of p62 in murine macrophages. Overexpression of p62 inhibited expression of multiple cytokines, IL-12p40, TNF-alpha, IL-1beta, IL-6, and IFN-beta, whereas p62 underexpression by small hairpin RNA markedly elevated their expression, indicating that p62 is a broad negative regulator of cytokine expression in stimulated macrophages. We show that p62 interacts with IFN regulatory factor 8 and Ro52, the transcription factor and ubiquitin E3 ligase that are important for IL-12p40 expression. This interaction, detectable at a late stage in stimulated macrophages, led to increased polyubiquitination and destabilization of IFN regulatory factor 8. We also show that upon macrophage stimulation, p62 binds to TNFR-associated factor 6, another E3 ligase important for NF-kappaB activation, but later this interaction was replaced by the recruitment of the deubiquitinating enzyme, cylindromatosis, an inhibitor of NF-kappaB activity. Recruitment of cylindromatosis coincided with reduced TNFR-associated factor 6 autoubiquitination and lower NF-kappaB activation. Our results indicate that p62 orchestrates orderly regulation of ubiquitin modification processes in macrophages to ensure attenuation of cytokine transcription postactivation. Together, p62 may provide a mechanism by which to control excessive inflammatory responses after macrophage activation.
Conflict of interest statement
The authors have no financial conflict of interest.
Figures



References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

