Sialylated multivalent antigens engage CD22 in trans and inhibit B cell activation
- PMID: 19202057
- PMCID: PMC2650292
- DOI: 10.1073/pnas.0807207106
Sialylated multivalent antigens engage CD22 in trans and inhibit B cell activation
Abstract
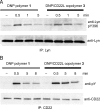
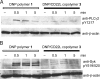
CD22 is an inhibitory coreceptor on the surface of B cells that attenuates B cell antigen receptor (BCR) signaling and, therefore, B cell activation. Elucidating the molecular mechanisms underlying the inhibitory activity of CD22 is complicated by the ubiquity of CD22 ligands. Although antigens can display CD22 ligands, the receptor is known to bind to sialylated glycoproteins on the cell surface. The propinquity of CD22 and cell-surface glycoprotein ligands has led to the conclusion that the inhibitory properties of the receptor are due to cis interactions. Here, we examine the functional consequences of trans interactions by employing sialylated multivalent antigens that can engage both CD22 and the BCR. Exposure of B cells to sialylated antigens results in the inhibition of key steps in BCR signaling. These results reveal that antigens bearing CD22 ligands are powerful suppressors of B cell activation. The ability of sialylated antigens to inhibit BCR signaling through trans CD22 interactions reveals a previously unrecognized role for the Siglec-family of receptors as modulators of immune signaling.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



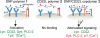
References
-
- Crocker PR, Paulson JC, Varki A. Siglecs and their roles in the immune system. Nat Rev Immunol. 2007;7:255. - PubMed
-
- Nitschke L. The role of CD22 and other inhibitory co-receptors in B-cell activation. Curr Opin Immunol. 2005;17:290–297. - PubMed
-
- O'Keefe TL, Williams GT, Davies SL, Neuberger MS. Hyperresponsive B cells in CD22-deficient mice. Science. 1996;274:798–801. - PubMed
-
- Gallo EM, Cante-Barrett K, Crabtree GR. Lymphocyte calcium signaling from membrane to nucleus. Nat Immunol. 2006;7:25–32. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

