Enhanced sensitivity to DSS colitis caused by a hypomorphic Mbtps1 mutation disrupting the ATF6-driven unfolded protein response
- PMID: 19202076
- PMCID: PMC2651297
- DOI: 10.1073/pnas.0813036106
Enhanced sensitivity to DSS colitis caused by a hypomorphic Mbtps1 mutation disrupting the ATF6-driven unfolded protein response
Abstract
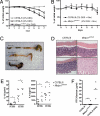
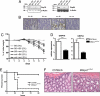
Here, we describe an N-ethyl-N-nitrosourea (ENU)-induced missense error in the membrane-bound transcription factor peptidase site 1 (S1P)-encoding gene (Mbtps1) that causes enhanced susceptibility to dextran sodium sulfate (DSS)-induced colitis. S1P cleaves and activates cAMP response element binding protein/ATF transcription factors, the sterol regulatory element-binding proteins (SREBPs), and other proteins of both endogenous and viral origin. Because S1P has a nonredundant function in the ATF6-dependent unfolded protein response (UPR), woodrat mice show diminished levels of major endoplasmic reticulum chaperones GRP78 (BiP) and GRP94 in the colon upon DSS administration. Experiments with bone marrow chimeric mice reveal a requirement for S1P in nonhematopoietic cells, without which a diminished UPR and colitis develop.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


References
-
- Ron D, Walter P. Signal integration in the endoplasmic reticulum unfolded protein response. Nat Rev Mol Cell Biol. 2007;8:519–529. - PubMed
-
- Todd DJ, Lee AH, Glimcher LH. The endoplasmic reticulum stress response in immunity and autoimmunity. Nat Rev Immunol. 2008;8:663–674. - PubMed
-
- Ye J, et al. ER stress induces cleavage of membrane-bound ATF6 by the same proteases that process SREBPs. Mol Cell. 2000;6:1355–1364. - PubMed
-
- Bertolotti A, Zhang Y, Hendershot LM, Harding HP, Ron D. Dynamic interaction of BiP and ER stress transducers in the unfolded-protein response. Nat Cell Biol. 2000;2:326–332. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

