AAA+ Ring and linker swing mechanism in the dynein motor
- PMID: 19203583
- PMCID: PMC2706395
- DOI: 10.1016/j.cell.2008.11.049
AAA+ Ring and linker swing mechanism in the dynein motor
Abstract
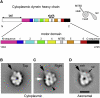
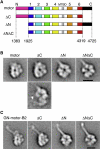
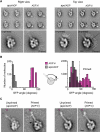
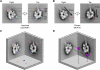
Dynein ATPases power diverse microtubule-based motilities. Each dynein motor domain comprises a ring-like head containing six AAA+ modules and N- and C-terminal regions, together with a stalk that binds microtubules. How these subdomains are arranged and generate force remains poorly understood. Here, using electron microscopy and image processing of tagged and truncated Dictyostelium cytoplasmic dynein constructs, we show that the heart of the motor is a hexameric ring of AAA+ modules, with the stalk emerging opposite the primary ATPase site (AAA1). The C-terminal region is not an integral part of the ring but spans between AAA6 and near the stalk base. The N-terminal region includes a lever-like linker whose N terminus swings by approximately 17 nm during the ATPase cycle between AAA2 and the stalk base. Together with evidence of stalk tilting, which may communicate changes in microtubule binding affinity, these findings suggest a model for dynein's structure and mechanism.
Figures



Comment in
-
Dynein swings into action.Cell. 2009 Feb 6;136(3):395-6. doi: 10.1016/j.cell.2009.01.026. Cell. 2009. PMID: 19203573
References
-
- Burgess S.A., Knight P.J. Is the dynein motor a winch? Curr. Opin. Struct. Biol. 2004;14:138–146. - PubMed
-
- Burgess S.A., Walker M.L., Sakakibara H., Knight P.J., Oiwa K. Dynein structure and power stroke. Nature. 2003;421:715–718. - PubMed
-
- Burgess S.A., Walker M.L., Thirumurugan K., Trinick J., Knight P.J. Use of negative stain and single-particle image processing to explore dynamic properties of flexible macromolecules. J. Struct. Biol. 2004;147:247–258. - PubMed
-
- Burgess S.A., Walker M.L., Sakakibara H., Oiwa K., Knight P.J. The structure of dynein-c by negative stain electron microscopy. J. Struct. Biol. 2004;146:205–216. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 080709/Z/06/Z/WT_/Wellcome Trust/United Kingdom
- BB/E00928X/1/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- WT_/Wellcome Trust/United Kingdom
- 24/B19478/BB_/Biotechnology and Biological Sciences Research Council/United Kingdom
- 078115/Z/05/Z/WT_/Wellcome Trust/United Kingdom
LinkOut - more resources
Full Text Sources
Other Literature Sources

