Effect of casopitant, a novel NK-1 antagonist, on the pharmacokinetics of dolasetron and granisetron
- PMID: 19205754
- PMCID: PMC2726911
- DOI: 10.1007/s00520-008-0572-4
Effect of casopitant, a novel NK-1 antagonist, on the pharmacokinetics of dolasetron and granisetron
Abstract
Objective: The objective of this study was to characterize the impact of casopitant, a novel neurokinin-1 receptor antagonist under investigation for the prevention of postoperative and chemotherapy-induced nausea and vomiting, on the pharmacokinetics of the commonly prescribed 5-hydroxytryptamine receptor 3 receptor antagonists, dolasetron or granisetron.
Materials and methods: In a phase I, open-label, two-part, two-period, single-sequence study, two cohorts of healthy subjects received either oral dolasetron (100 mg once daily for 3 days) or oral granisetron (2 mg once daily for 3 days) alone (period 1) and combined with oral casopitant, 150 mg day 1, 50 mg days 2 and 3 (period 2). Pharmacokinetics of hydrodolasetron and granisetron were assessed on days 1 and 3 of each period. Log-transformed area under the curve (AUC) and Cmax were statistically analyzed by performing an analysis of variance. Eighteen subjects were enrolled in the dolasetron cohort; nine subjects were CYP2D6 extensive metabolizers (EMs) and nine subjects were CYP2D6 poor metabolizers. Nineteen subjects were enrolled in the granisetron cohort.
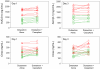
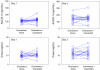
Results: The largest changes in hydrodolasetron exposure after coadministration with casopitant were seen in CYP2D6 EMs, with a 24% increase in hydrodolasetron AUC on day 1 and 30% increase in Cmax on days 1 and 3. All other changes in hydrodolasetron exposure were <20%, and granisetron exposure was not altered to any relevant extent (<11%).
Conclusion: None of the changes observed are considered clinically meaningful, and coadministration of casopitant with dolasetron or granisetron was well tolerated.
Figures
References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1097/00000542-199909000-00022', 'is_inner': False, 'url': 'https://doi.org/10.1097/00000542-199909000-00022'}, {'type': 'PubMed', 'value': '10485781', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/10485781/'}]}
- Apfel CC, Laara E, Koivuranta M et al (1999) A simplified risk score for predicting postoperative nausea and vomiting. Anesthesiology 91:693–700. doi:10.1097/00000542-199909000-00022 - PubMed
-
- Aventis (2006) Anzemet prescribing information. June
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1002/ddr.430340306', 'is_inner': False, 'url': 'https://doi.org/10.1002/ddr.430340306'}]}
- Bigaud M, Elands J, Kastner PR et al (1995) Pharmacology of the human metabolites of dolasetron, an antiemetic 5-HT3 receptor antagonist. Drug Dev Res 34:289–296. doi:10.1002/ddr.430340306
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'PMC', 'value': 'PMC1364919', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC1364919/'}, {'type': 'PubMed', 'value': '7888294', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/7888294/'}]}
- Bloomer JC, Baldwin SJ, Smith GJ et al (1994) Characterization of the cytochrome P450 enzymes involved in the in vitro metabolism of granisetron. Br J Clin Pharmacol 38:557–566 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1517/14622416.3.2.229', 'is_inner': False, 'url': 'https://doi.org/10.1517/14622416.3.2.229'}, {'type': 'PubMed', 'value': '11972444', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/11972444/'}]}
- Bradford LD (2002) CYP2D6 allele frequency in European Caucasians, Asians, Africans, and their descendants. Pharmacogenomics 3:229–243. doi:10.1517/14622416.3.2.229 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

