Inhibition of contraction-stimulated AMP-activated protein kinase inhibits contraction-stimulated increases in PAS-TBC1D1 and glucose transport without altering PAS-AS160 in rat skeletal muscle
- PMID: 19208911
- PMCID: PMC2671042
- DOI: 10.2337/db08-1477
Inhibition of contraction-stimulated AMP-activated protein kinase inhibits contraction-stimulated increases in PAS-TBC1D1 and glucose transport without altering PAS-AS160 in rat skeletal muscle
Abstract
Objective: Phosphorylation of two members of the TBC1 domain family of proteins, Akt substrate of 160 kDa (AS160, also known as TBC1D4) and TBC1D1, has been implicated in the regulation of glucose transport in skeletal muscle. Insulin-stimulated phosphorylation (measured using the phospho-Akt substrate [PAS] antibody) of AS160 and TBC1D1 appears to occur in an Akt-dependent manner, but the kinases responsible for contraction-stimulated PAS-AS160 and PAS-TBC1D1 remain unclear. AMP-activated protein kinase (AMPK) and Akt, both activated by contraction, can each phosphorylate AS160 and TBC1D1 in cell-free assays.
Research design and methods: To evaluate the roles of AMPK and Akt on insulin- or contraction-stimulated PAS-AS160, PAS-TBC1D1, and glucose transport, rat epitrochlearis was incubated with and without compound C (inhibitor of AMPK) or Wortmannin (inhibitor of phosphatidylinositol [PI] 3-kinase, which is upstream of Akt) before and during insulin stimulation or contraction.
Results: Insulin-stimulated glucose transport and phosphorylation of both AS160 and TBC1D1 were completely inhibited by Wortmannin. Wortmannin eliminated contraction stimulation of phospho-Ser(21/9)glycogen synthase kinase 3alpha/beta (pGSK3; Akt substrate) and PAS-AS160 but did not significantly alter pAMPK, phospho-Ser79acetyl CoA carboxylase (pACC; AMPK substrate), PAS-TBC1D1, or glucose transport in contraction-stimulated muscle. Compound C completely inhibited contraction-stimulated pACC and PAS-TBC1D1 and partially blocked glucose transport, but it did not significantly alter pAkt, pGSK3, or PAS-AS160.
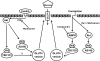
Conclusions: These data suggest that 1) insulin stimulates glucose transport and phosphorylation of AS160 and TBC1D1 in a PI 3-kinase/Akt-dependent manner, 2) contraction stimulates PAS-AS160 (but not PAS-TBC1D1 or glucose transport) in a PI 3-kinase/Akt-dependent manner, and 3) contraction stimulates PAS-TBC1D1 and glucose transport (but not PAS-AS160) in an AMPK-dependent manner.
Figures




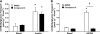
Similar articles
-
Roles of TBC1D1 and TBC1D4 in insulin- and exercise-stimulated glucose transport of skeletal muscle.Diabetologia. 2015 Jan;58(1):19-30. doi: 10.1007/s00125-014-3395-5. Epub 2014 Oct 4. Diabetologia. 2015. PMID: 25280670 Free PMC article. Review.
-
Contraction-stimulated glucose transport in rat skeletal muscle is sustained despite reversal of increased PAS-phosphorylation of AS160 and TBC1D1.J Appl Physiol (1985). 2008 Dec;105(6):1788-95. doi: 10.1152/japplphysiol.90838.2008. Epub 2008 Sep 25. J Appl Physiol (1985). 2008. PMID: 18818383 Free PMC article.
-
Discovery of TBC1D1 as an insulin-, AICAR-, and contraction-stimulated signaling nexus in mouse skeletal muscle.J Biol Chem. 2008 Apr 11;283(15):9787-96. doi: 10.1074/jbc.M708839200. Epub 2008 Feb 13. J Biol Chem. 2008. PMID: 18276596 Free PMC article.
-
Thr649Ala-AS160 knock-in mutation does not impair contraction/AICAR-induced glucose transport in mouse muscle.Am J Physiol Endocrinol Metab. 2012 May 15;302(9):E1036-43. doi: 10.1152/ajpendo.00379.2011. Epub 2012 Feb 7. Am J Physiol Endocrinol Metab. 2012. PMID: 22318952 Free PMC article.
-
Role of Akt substrate of 160 kDa in insulin-stimulated and contraction-stimulated glucose transport.Appl Physiol Nutr Metab. 2007 Jun;32(3):557-66. doi: 10.1139/H07-026. Appl Physiol Nutr Metab. 2007. PMID: 17510697 Review.
Cited by
-
Roles of TBC1D1 and TBC1D4 in insulin- and exercise-stimulated glucose transport of skeletal muscle.Diabetologia. 2015 Jan;58(1):19-30. doi: 10.1007/s00125-014-3395-5. Epub 2014 Oct 4. Diabetologia. 2015. PMID: 25280670 Free PMC article. Review.
-
17-β estradiol attenuates ovariectomy-induced changes in cardiomyocyte contractile function via activation of AMP-activated protein kinase.Toxicol Lett. 2015 Jan 5;232(1):253-62. doi: 10.1016/j.toxlet.2014.11.012. Epub 2014 Nov 13. Toxicol Lett. 2015. PMID: 25448287 Free PMC article.
-
Elevation of muscle temperature stimulates muscle glucose uptake in vivo and in vitro.J Physiol Sci. 2013 Nov;63(6):409-18. doi: 10.1007/s12576-013-0278-3. Epub 2013 Jul 9. J Physiol Sci. 2013. PMID: 23836025 Free PMC article.
-
Contraction regulates site-specific phosphorylation of TBC1D1 in skeletal muscle.Biochem J. 2010 Oct 15;431(2):311-20. doi: 10.1042/BJ20101100. Biochem J. 2010. PMID: 20701589 Free PMC article.
-
Identification and Association of SNPs in TBC1D1 Gene with Growth Traits in Two Rabbit Breeds.Asian-Australas J Anim Sci. 2013 Nov;26(11):1529-35. doi: 10.5713/ajas.2013.13278. Asian-Australas J Anim Sci. 2013. PMID: 25049738 Free PMC article.
References
-
- Douen AG, Ramlal T, Rastogi S, Bilan PJ, Cartee GD, Vranic M, Holloszy JO, Klip A: Exercise induces recruitment of the “insulin-responsive glucose transporter”: evidence for distinct intracellular insulin- and exercise-recruitable transporter pools in skeletal muscle. J Biol Chem 1990; 265: 13427– 13430 - PubMed
-
- Coderre L, Kandror KV, Vallega G, Pilch PF: Identification and characterization of an exercise-sensitive pool of glucose transporters in skeletal muscle. J Biol Chem 1995; 270: 27584– 27588 - PubMed
-
- Cartee GD, Wojtaszewski JF: Role of Akt substrate of 160 kDa in insulin-stimulated and contraction-stimulated glucose transport. Appl Physiol Nutr Metab 2007; 32: 557– 566 - PubMed
-
- Hayashi T, Hirshman MF, Kurth EJ, Winder WW, Goodyear LJ: Evidence for 5′ AMP-activated protein kinase mediation of the effect of muscle contraction on glucose transport. Diabetes 1998; 47: 1369– 1373 - PubMed
-
- Cho H, Mu J, Kim JK, Thorvaldsen JL, Chu Q, Crenshaw EB, 3rd, Kaestner KH, Bartolomei MS, Shulman GI, Birnbaum MJ: Insulin resistance and a diabetes mellitus-like syndrome in mice lacking the protein kinase Akt2 (PKB beta). Science 2001; 292: 1728– 1731 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

