Autocrine regulation of T-cell activation by ATP release and P2X7 receptors
- PMID: 19211924
- PMCID: PMC2718802
- DOI: 10.1096/fj.08-126458
Autocrine regulation of T-cell activation by ATP release and P2X7 receptors
Abstract
T-cell activation requires the influx of extracellular calcium, although mechanistic details regarding such activation are not fully defined. Here, we show that P2X(7) receptors play a key role in calcium influx and downstream signaling events associated with the activation of T cells. By real-time PCR and immunohistochemistry, we find that Jurkat T cells and human CD4(+) T cells express abundant P2X(7) receptors. We show, using a novel fluorescent microscopy technique, that T-cell receptor (TCR) stimulation triggers the rapid release of ATP (<100 microM). This release of ATP is required for TCR-mediated calcium influx, NFAT activation, and interleukin-2 (IL-2) production. TCR activation up-regulates P2X(7) receptor gene expression. Removal of extracellular ATP by apyrase or alkaline phosphatase treatment, inhibition of ATP release with the maxi-anion channel blocker gadolinium chloride, or siRNA silencing of P2X(7) receptors blocks calcium entry and inhibits T-cell activation. Moreover, lymphocyte activation is impaired in C57BL/6 mice that express poorly functional P2X(7) receptors, compared to control BALB/c mice, which express fully functional P2X(7) receptors. We conclude that ATP release and autocrine, positive feedback through P2X(7) receptors is required for the effective activation of T cells.
Figures

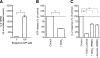
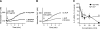
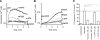



References
-
- Zeyda M, Stulnig T M. Lipid Rafts & Co.: an integrated model of membrane organization in T cell activation. Prog Lipid Res. 2006;45:187–202. - PubMed
-
- Hogan P G, Chen L, Nardone J, Rao A. Transcriptional regulation by calcium, calcineurin, and NFAT. Genes Dev. 2003;17:2205–2232. - PubMed
-
- Yip L, Cheung C W, Corriden R, Chen Y, Insel P A, Junger W G. Hypertonic stress regulates T-cell function by the opposing actions of extracellular adenosine triphosphate and adenosine. Shock. 2007;27:242–250. - PubMed
-
- Loomis W H, Namiki S, Ostrom R S, Insel P A, Junger W G. Hypertonic stress increases T cell interleukin-2 expression through a mechanism that involves ATP release, P2 receptor, and p38 MAPK activation. J Biol Chem. 2003;278:4590–4596. - PubMed
-
- Corriden R, Insel P A, Junger W G. A novel method using fluorescence microscopy for real-time assessment of ATP release from individual cells. Am J Physiol Cell Physiol. 2007;293:C1420–C1425. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

