The fragile X mental retardation protein in circadian rhythmicity and memory consolidation
- PMID: 19214804
- PMCID: PMC3848790
- DOI: 10.1007/s12035-009-8057-0
The fragile X mental retardation protein in circadian rhythmicity and memory consolidation
Abstract
The control of new protein synthesis provides a means to locally regulate the availability of synaptic components necessary for dynamic neuronal processes. The fragile X mental retardation protein (FMRP), an RNA-binding translational regulator, is a key player mediating appropriate synaptic protein synthesis in response to neuronal activity levels. Loss of FMRP causes fragile X syndrome (FraX), the most commonly inherited form of mental retardation and autism spectrum disorders. FraX-associated translational dysregulation causes wide-ranging neurological deficits including severe impairments of biological rhythms, learning processes, and memory consolidation. Dysfunction in cytoskeletal regulation and synaptic scaffolding disrupts neuronal architecture and functional synaptic connectivity. The understanding of this devastating disease and the implementation of meaningful treatment strategies require a thorough exploration of the temporal and spatial requirements for FMRP in establishing and maintaining neural circuit function.
Figures


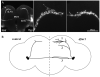
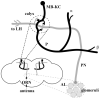

References
-
- Verkerk AJ, Pieretti M, Sutcliffe JS, Fu YH, Kuhl DP, Pizzuti A, Reiner O, Richards S, Victoria MF, Zhang FP, et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell. 1991;65:905–914. - PubMed
-
- Skinner M, Hooper S, Hatton DD, Roberts J, Mirrett P, Schaaf J, Sullivan K, Wheeler A, Bailey DB., Jr. Mapping nonverbal IQ in young boys with fragile X syndrome. Am J Med Genet A. 2005;132:25–32. - PubMed
-
- Cohen D, Pichard N, Tordjman S, Baumann C, Burglen L, Excoffier E, Lazar G, Mazet P, Pinquier C, Verloes A, Heron D. Specific genetic disorders and autism: clinical contribution towards their identification. J Autism Dev Disord. 2005;35:103–116. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

