Synergistic and antagonistic interactions between tetrodotoxin and mu-conotoxin in blocking voltage-gated sodium channels
- PMID: 19221510
- PMCID: PMC2878737
- DOI: 10.4161/chan.3.1.7500
Synergistic and antagonistic interactions between tetrodotoxin and mu-conotoxin in blocking voltage-gated sodium channels
Abstract
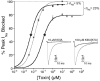
Tetrodotoxin (TTX) is the quintessential ligand of voltage-gated sodium channels (NaVs). Like TTX, mu-conotoxin peptides are pore blockers, and both toxins have helped to define the properties of neurotoxin receptor Site 1 of NaVs. Here, we report unexpected results showing that the recently discovered mu-conotoxin KIIIA and TTX can simultaneously bind to Site 1 and act in concert. Results with saturating concentrations of peptide applied to voltage-clamped Xenopus oocytes expressing brain NaV1.2, and single-channel recordings from brain channels in lipid bilayers, show that KIIIA or its analog, KIIIA[K7A], block partially, with a residual current that can be completely blocked by TTX. In addition, the kinetics of block by TTX and peptide are each affected by the prior presence of the other toxin. For example, bound peptide slows subsequent binding of TTX (an antagonistic interaction) and slows TTX dissociation when both toxins are bound (a synergistic effect on block). The overall functional consequence resulting from the combined action of the toxins depends on the quantitative balance between these opposing actions. The results lead us to postulate that in the bi-liganded NaV complex, TTX is bound between the peptide and the selectivity filter. These observations refine our view of Site 1 and open new possibilities in NaV pharmacology.
Figures


Similar articles
-
Cooccupancy of the outer vestibule of voltage-gated sodium channels by micro-conotoxin KIIIA and saxitoxin or tetrodotoxin.J Neurophysiol. 2010 Jul;104(1):88-97. doi: 10.1152/jn.00145.2010. Epub 2010 Apr 21. J Neurophysiol. 2010. PMID: 20410356 Free PMC article.
-
μ-conotoxin KIIIA derivatives with divergent affinities versus efficacies in blocking voltage-gated sodium channels.Biochemistry. 2010 Jun 15;49(23):4804-12. doi: 10.1021/bi100207k. Biochemistry. 2010. PMID: 20459109 Free PMC article.
-
The TTX metabolite 4,9-anhydro-TTX is a highly specific blocker of the Na(v1.6) voltage-dependent sodium channel.Am J Physiol Cell Physiol. 2007 Aug;293(2):C783-9. doi: 10.1152/ajpcell.00070.2007. Epub 2007 May 23. Am J Physiol Cell Physiol. 2007. PMID: 17522141
-
The tetrodotoxin receptor of voltage-gated sodium channels--perspectives from interactions with micro-conotoxins.Mar Drugs. 2010 Jul 13;8(7):2153-61. doi: 10.3390/md8072153. Mar Drugs. 2010. PMID: 20714429 Free PMC article. Review.
-
Interaction between voltage-gated sodium channels and the neurotoxin, tetrodotoxin.Channels (Austin). 2008 Nov-Dec;2(6):407-12. doi: 10.4161/chan.2.6.7429. Epub 2008 Nov 12. Channels (Austin). 2008. PMID: 19098433 Review.
Cited by
-
Computational Design of High-Affinity Blockers for Sodium Channel NaV1.2 from μ-Conotoxin KIIIA.Mar Drugs. 2022 Feb 21;20(2):154. doi: 10.3390/md20020154. Mar Drugs. 2022. PMID: 35200683 Free PMC article.
-
A disulfide tether stabilizes the block of sodium channels by the conotoxin μO§-GVIIJ.Proc Natl Acad Sci U S A. 2014 Feb 18;111(7):2758-63. doi: 10.1073/pnas.1324189111. Epub 2014 Feb 4. Proc Natl Acad Sci U S A. 2014. PMID: 24497506 Free PMC article.
-
Co-expression of Na(V)β subunits alters the kinetics of inhibition of voltage-gated sodium channels by pore-blocking μ-conotoxins.Br J Pharmacol. 2013 Apr;168(7):1597-610. doi: 10.1111/bph.12051. Br J Pharmacol. 2013. PMID: 23146020 Free PMC article.
-
Cooccupancy of the outer vestibule of voltage-gated sodium channels by micro-conotoxin KIIIA and saxitoxin or tetrodotoxin.J Neurophysiol. 2010 Jul;104(1):88-97. doi: 10.1152/jn.00145.2010. Epub 2010 Apr 21. J Neurophysiol. 2010. PMID: 20410356 Free PMC article.
-
μ-conotoxin KIIIA derivatives with divergent affinities versus efficacies in blocking voltage-gated sodium channels.Biochemistry. 2010 Jun 15;49(23):4804-12. doi: 10.1021/bi100207k. Biochemistry. 2010. PMID: 20459109 Free PMC article.
References
-
- Catterall WA, Goldin AL, Waxman SG. International Union of Pharmacology. XLVII. Nomenclature and structure-function relationships of voltage-gated sodium channels. Pharmacological reviews. 2005;57:397–409. - PubMed
-
- French RJ, Terlau H. Sodium channel toxins--receptor targeting and therapeutic potential. Curr Med Chem. 2004;11:3053–64. - PubMed
-
- Wood JN, Boorman JP, Okuse K, Baker MD. Voltage-gated sodium channels and pain pathways. Journal of neurobiology. 2004;61:55–71. - PubMed
-
- Cestele S, Catterall WA. Molecular mechanisms of neurotoxin action on voltage-gated sodium channels. Biochimie. 2000;82:883–92. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
