Biochemical, structural, and biomarker evidence for calpain-mediated cytoskeletal change after diffuse brain injury uncomplicated by contusion
- PMID: 19225412
- PMCID: PMC2779123
- DOI: 10.1097/NEN.0b013e3181996bfe
Biochemical, structural, and biomarker evidence for calpain-mediated cytoskeletal change after diffuse brain injury uncomplicated by contusion
Abstract
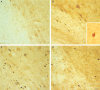
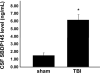
Calpain-mediated degradation of the cytoskeletal protein alpha-II-spectrin has been implicated in the pathobiology of experimental and human traumatic brain injury (TBI). Spectrin proteolysis after diffuse/widespread TBI uncomplicated by either subtle or overt contusion and/or mass lesions, (i.e. mild to moderate TBI), has not been previously evaluated. To determine the spatiotemporal pattern and cellular localization of calpain-mediated spectrin proteolysis after diffuse/widespread TBI and the extent to which parenchymal changes in calpain-mediated spectrin proteolysis are reflected in the cerebrospinal fluid, adult rats were subjected to a moderate midline fluid percussion injury and allowed to survive for 3 hours to 7 days postinjury. Light and electron microscopic immunocytochemical and Western blot analyses were performed to identify the calpain-specific 145-kDa breakdown product of alpha-II-spectrin (SBDP145). After diffuse TBI, enhanced levels of SBDP145 immunoreactivity were observed in the neocortex, subcortical white matter, thalamus, and hippocampus, peaking between 24 and 48 hours postinjury. Immunoreactivity was localized almost exclusively to damaged axons and axonal terminal debris. Heightened levels of SBDP145 were also observed in the cerebrospinal fluid at 24 hours postinjury. These results confirm the widespread occurrence of calpain-mediated spectrin proteolysis after diffuse TBI without contusion and support the potential utility of SBDPs as biomarkers of a diffusely injured brain.
Figures





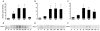
Similar articles
-
Accumulation of non-erythroid alpha II-spectrin and calpain-cleaved alpha II-spectrin breakdown products in cerebrospinal fluid after traumatic brain injury in rats.J Neurochem. 2001 Sep;78(6):1297-306. doi: 10.1046/j.1471-4159.2001.00510.x. J Neurochem. 2001. PMID: 11579138
-
Regional calpain and caspase-3 proteolysis of alpha-spectrin after traumatic brain injury.Neuroreport. 1998 Aug 3;9(11):2437-42. doi: 10.1097/00001756-199808030-00002. Neuroreport. 1998. PMID: 9721910
-
The role of calpain-mediated spectrin proteolysis in traumatically induced axonal injury.J Neuropathol Exp Neurol. 1999 Apr;58(4):365-75. doi: 10.1097/00005072-199904000-00007. J Neuropathol Exp Neurol. 1999. PMID: 10218632
-
Spectrin and calpain: a 'target' and a 'sniper' in the pathology of neuronal cells.Cell Mol Life Sci. 2005 Sep;62(17):1913-24. doi: 10.1007/s00018-005-5097-0. Cell Mol Life Sci. 2005. PMID: 15990959 Free PMC article. Review.
-
Calpain as a therapeutic target in traumatic brain injury.Neurotherapeutics. 2010 Jan;7(1):31-42. doi: 10.1016/j.nurt.2009.11.002. Neurotherapeutics. 2010. PMID: 20129495 Free PMC article. Review.
Cited by
-
Clinical characteristics and pathophysiological mechanisms of focal and diffuse traumatic brain injury.J Cell Mol Med. 2010 Oct;14(10):2381-92. doi: 10.1111/j.1582-4934.2010.01164.x. J Cell Mol Med. 2010. PMID: 20738443 Free PMC article. Review.
-
Recovery of neurological function despite immediate sleep disruption following diffuse brain injury in the mouse: clinical relevance to medically untreated concussion.Sleep. 2014 Apr 1;37(4):743-52. doi: 10.5665/sleep.3582. Sleep. 2014. PMID: 24899763 Free PMC article.
-
Mechanosensation in traumatic brain injury.Neurobiol Dis. 2021 Jan;148:105210. doi: 10.1016/j.nbd.2020.105210. Epub 2020 Nov 28. Neurobiol Dis. 2021. PMID: 33259894 Free PMC article. Review.
-
Post-injury administration of the mitochondrial permeability transition pore inhibitor, NIM811, is neuroprotective and improves cognition after traumatic brain injury in rats.J Neurotrauma. 2011 Sep;28(9):1845-53. doi: 10.1089/neu.2011.1755. Epub 2011 Aug 29. J Neurotrauma. 2011. PMID: 21875332 Free PMC article.
-
Biomarkers improve clinical outcome predictors of mortality following non-penetrating severe traumatic brain injury.Neurocrit Care. 2015 Feb;22(1):52-64. doi: 10.1007/s12028-014-0028-2. Neurocrit Care. 2015. PMID: 25052159
References
-
- Langlois JA, Rutland-Brown W, Wald MM. The epidemiology and impact of traumatic brain injury: A brief overview. J Head Trauma Rehabil. 2006;21:375–78. - PubMed
-
- Bramlett HM, Dietrich WD. Progressive damage after brain and spinal cord injury: Pathomechanisms and treatment strategies. Prog Brain Res. 2007;161:125–41. - PubMed
-
- Buki A, Povlishock JT. All roads lead to disconnection?--Traumatic axonal injury revisited. Acta Neurochir (Wien) 2006;148:181–93. - PubMed
-
- Farkas O, Povlishock JT. Cellular and subcellular change evoked by diffuse traumatic brain injury: A complex web of change extending far beyond focal damage. Prog Brain Res. 2007;161:43–59. - PubMed
-
- Povlishock JT, Katz DI. Update of neuropathology and neurological recovery after traumatic brain injury. J Head Trauma Rehabil. 2005;20:76–94. - PubMed

