Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient
- PMID: 19226183
- PMCID: PMC2642879
- DOI: 10.1371/journal.pmed.1000029
Donor-derived brain tumor following neural stem cell transplantation in an ataxia telangiectasia patient
Abstract
Background: Neural stem cells are currently being investigated as potential therapies for neurodegenerative diseases, stroke, and trauma. However, concerns have been raised over the safety of this experimental therapeutic approach, including, for example, whether there is the potential for tumors to develop from transplanted stem cells.
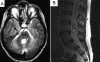
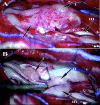
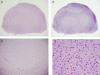
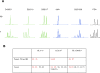
Methods and findings: A boy with ataxia telangiectasia (AT) was treated with intracerebellar and intrathecal injection of human fetal neural stem cells. Four years after the first treatment he was diagnosed with a multifocal brain tumor. The biopsied tumor was diagnosed as a glioneuronal neoplasm. We compared the tumor cells and the patient's peripheral blood cells by fluorescent in situ hybridization using X and Y chromosome probes, by PCR for the amelogenin gene X- and Y-specific alleles, by MassArray for the ATM patient specific mutation and for several SNPs, by PCR for polymorphic microsatellites, and by human leukocyte antigen (HLA) typing. Molecular and cytogenetic studies showed that the tumor was of nonhost origin suggesting it was derived from the transplanted neural stem cells. Microsatellite and HLA analysis demonstrated that the tumor is derived from at least two donors.
Conclusions: This is the first report of a human brain tumor complicating neural stem cell therapy. The findings here suggest that neuronal stem/progenitor cells may be involved in gliomagenesis and provide the first example of a donor-derived brain tumor. Further work is urgently needed to assess the safety of these therapies.
Conflict of interest statement
Figures




References
-
- Lindvall O, Kokaia Z, Martinez-Serrano A. Stem cell therapy for human neurodegenerative disorders-how to make it work. Nat Med. 2004;10(Suppl):S42–50. - PubMed
-
- Lindvall O, Kokaia Z. Stem cells for the treatment of neurological disorders. Nature. 2006;441:1094–1096. - PubMed
-
- Flax JD, Aurora S, Yang C, Simonin C, Wills AM, et al. Engraftable human neural stem cells respond to developmental cues, replace neurons, and express foreign genes. Nat Biotechnol. 1998;16:1033–1039. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

