Anti-atherogenic effect of statins: role of nitric oxide, peroxynitrite and haem oxygenase-1
- PMID: 19226281
- PMCID: PMC2697732
- DOI: 10.1111/j.1476-5381.2009.00125.x
Anti-atherogenic effect of statins: role of nitric oxide, peroxynitrite and haem oxygenase-1
Abstract
Background and purpose: The pleiotropic effects of HMG-CoA inhibitors (statins), which include anti-inflammation, antioxidation and immunomodulation, are not yet fully understood. The present study was designed to elucidate the role of nitric oxide (NO), peroxynitrite (ONOO(-)) and haem oxygenase-1 (HO-1) in the anti-atherogenic effect of statins.
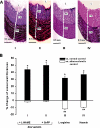
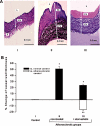
Experimental approach: Normal and atherosclerotic New Zealand rabbits were treated with atorvastatin or simvastatin in the presence or absence of inhibitors and promoters of endothelial nitric oxide synthase (eNOS) and HO-1. NO and ONOO(-) released from isolated aortae by calcium ionophore were measured with nanosensors placed 6 +/- 2 nm from aortic endothelium. Expression of eNOS and HO-1 protein, HO activity, plasma malondialdehyde (MDA) and vessel wall thickness were also measured.
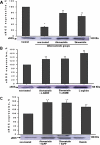
Key results: Hypercholesterolaemia decreased eNOS expression by 31 +/- 3%, decreased NO (230 +/- 16 vs. 433 +/- 17 nmol x L(-1) control) and increased cytotoxic ONOO(-) (299 +/- 15 vs. 187 +/- 11 nmol x L(-1) control). The concentration ratio of [NO]/[ONOO(-)] decreased from 2.3 +/- 0.1 (normal) to 0.7 +/- 0.1 indicating an increase of nitroxidative stress in atherosclerotic endothelium. Expression of HO-1 protein increased by 20 +/- 8% in atherosclerosis and further increased (about 30%) after treatment with statins. Statins partially restored the [NO]/[ONOO(-)] balance (1.5 +/- 0.1 for atorvastatin and 1.4 +/- 0.1 simvastatin), decreased MDA and wall thickening. Promoters of eNOS and HO-1 (L-arginine and haemin) ameliorated the [NO]/[ONOO(-)] ratio while their inhibitors (L-NAME or tin-protoporphyrin) showed no improvement in these ratio.
Conclusions and implications: Atherosclerosis induced an endothelial [NO]/[ONOO(-)] balance indicative of endothelial dysfunction. Statins showed anti-atherosclerotic effects mediated by HO-1/eNOS, restoring the [NO]/[ONOO(-)] imbalance and reducing lipid peroxidation.
Figures





Similar articles
-
Adverse balance of nitric oxide/peroxynitrite in the dysfunctional endothelium can be reversed by statins.J Cardiovasc Pharmacol. 2007 Oct;50(4):391-8. doi: 10.1097/FJC.0b013e31811f3fd0. J Cardiovasc Pharmacol. 2007. PMID: 18049306
-
Leptin-induced endothelial dysfunction in obesity.Am J Physiol Heart Circ Physiol. 2008 Oct;295(4):H1514-21. doi: 10.1152/ajpheart.00479.2008. Epub 2008 Aug 8. Am J Physiol Heart Circ Physiol. 2008. PMID: 18689498 Free PMC article.
-
Synergistic effect of amlodipine and atorvastatin in reversing LDL-induced endothelial dysfunction.Pharm Res. 2008 Aug;25(8):1798-806. doi: 10.1007/s11095-007-9491-1. Epub 2007 Dec 18. Pharm Res. 2008. PMID: 18087679
-
Advances in the molecular mechanisms of statins in regulating endothelial nitric oxide bioavailability: Interlocking biology between eNOS activity and L-arginine metabolism.Biomed Pharmacother. 2024 Feb;171:116192. doi: 10.1016/j.biopha.2024.116192. Epub 2024 Jan 22. Biomed Pharmacother. 2024. PMID: 38262153 Review.
-
Therapeutic effect of enhancing endothelial nitric oxide synthase (eNOS) expression and preventing eNOS uncoupling.Br J Pharmacol. 2011 Sep;164(2):213-23. doi: 10.1111/j.1476-5381.2010.01196.x. Br J Pharmacol. 2011. PMID: 21198553 Free PMC article. Review.
Cited by
-
Therapeutic Efficacy of Antioxidants in Ameliorating Obesity Phenotype and Associated Comorbidities.Front Pharmacol. 2020 Aug 13;11:1234. doi: 10.3389/fphar.2020.01234. eCollection 2020. Front Pharmacol. 2020. PMID: 32903449 Free PMC article. Review.
-
Association of Circulating Heme Oxygenase-1, Lipid Profile and Coronary Disease Phenotype in Patients with Chronic Coronary Syndrome.Antioxidants (Basel). 2021 Dec 15;10(12):2002. doi: 10.3390/antiox10122002. Antioxidants (Basel). 2021. PMID: 34943105 Free PMC article.
-
Heme oxygenase-1 in inflammation and cardiovascular disease.Am J Cardiovasc Dis. 2011;1(2):150-8. Epub 2011 Jul 28. Am J Cardiovasc Dis. 2011. PMID: 22254194 Free PMC article.
-
Antioxidation Effect of Simvastatin in Aorta and Hippocampus: A Rabbit Model Fed High-Cholesterol Diet.Oxid Med Cell Longev. 2016;2016:6929306. doi: 10.1155/2016/6929306. Epub 2015 Dec 20. Oxid Med Cell Longev. 2016. PMID: 26798426 Free PMC article.
-
Interdependence of Anti-Inflammatory and Antioxidant Properties of Squalene-Implication for Cardiovascular Health.Life (Basel). 2021 Jan 29;11(2):103. doi: 10.3390/life11020103. Life (Basel). 2021. PMID: 33573041 Free PMC article. Review.
References
-
- Ahmed H, McLaughlin BE, Soong J, Marks GS, Brien JF, Nakatsu K. The source of endogenous carbon monoxide formation in human placental chorionic villi. Cell Mol Biol (Noisy-le-grand. 2005;51:447–451. - PubMed
-
- Beckman JSWDA, Crow JP. Methods in Nitric Oxide. New York: John Wiley and Sons, Ltd; 1996.
-
- Cyrus T, Tang LX, Rokach J, FitzGerald GA, Pratico D. Lipid peroxidation and platelet activation in murine atherosclerosis. Circulation. 2001;104:1940–1945. - PubMed
-
- Durante W, Kroll MH, Christodoulides N, Peyton KJ, Schafer AI. Nitric oxide induces heme oxygenase-1 gene expression and carbon monoxide production in vascular smooth muscle cells. Circ Res. 1997;80:557–564. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

