Oligomeric amyloid beta associates with postsynaptic densities and correlates with excitatory synapse loss near senile plaques
- PMID: 19228947
- PMCID: PMC2656196
- DOI: 10.1073/pnas.0811698106
Oligomeric amyloid beta associates with postsynaptic densities and correlates with excitatory synapse loss near senile plaques
Abstract
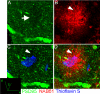
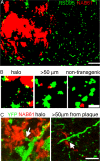
Synapse loss correlates with a cognitive decline in Alzheimer's disease (AD), but whether this is caused by fibrillar deposits known as senile plaques or soluble oligomeric forms of amyloid beta (Abeta) is controversial. By using array tomography, a technique that combines ultrathin sectioning of tissue with immunofluorescence, allowing precise quantification of small structures, such as synapses, we have tested the hypothesis that oligomeric Abeta surrounding plaques contributes to synapse loss in a mouse model of AD. We find that senile plaques are surrounded by a halo of oligomeric Abeta. Analysis of >14,000 synapses (represented by PSD95-stained excitatory synapses) shows that there is a 60% loss of excitatory synapses in the halo of oligomeric Abeta surrounding plaques and that the density increases to reach almost control levels in volumes further than 50 microm from a plaque in an approximately linear fashion (linear regression, r(2) = 0.9; P < 0.0001). Further, in transgenic cortex, microdeposits of oligomeric Abeta associate with a subset of excitatory synapses, which are significantly smaller than those not in contact with oligomeric Abeta. The proportion of excitatory synapses associated with Abeta correlates with decreasing density (correlation, -0.588; P < 0.0001). These data show that senile plaques are a potential reservoir of oligomeric Abeta, which colocalizes with the postsynaptic density and is associated with spine collapse, reconciling the apparently competing schools of thought of "plaque" vs. "oligomeric Abeta" as the synaptotoxic species in the brain of AD patients.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Terry RD, et al. Physical basis of cognitive alterations in Alzheimer's disease: Synapse loss is the major correlate of cognitive impairment. Ann Neurol. 1991;30:572–580. - PubMed
-
- DeKosky ST, Scheff SW, Styren SD. Structural correlates of cognition in dementia: Quantification and assessment of synapse change. Neurodegeneration. 1996;5:417–421. - PubMed
-
- Coleman PD, Yao PJ. Synaptic slaughter in Alzheimer's disease. Neurobiol Aging. 2003;24:1023–1027. - PubMed
-
- Snyder EM, et al. Regulation of NMDA receptor trafficking by amyloid-β. Nat Neurosci. 2005;8:1051–1058. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

