Development of spontaneous recurrent seizures after kainate-induced status epilepticus
- PMID: 19228963
- PMCID: PMC2897752
- DOI: 10.1523/JNEUROSCI.0980-08.2009
Development of spontaneous recurrent seizures after kainate-induced status epilepticus
Abstract
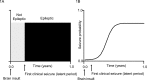
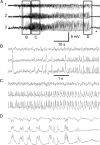
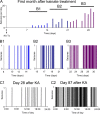
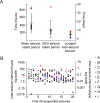
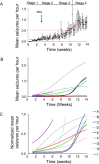
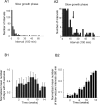
Acquired epilepsy (i.e., after an insult to the brain) is often considered to be a progressive disorder, and the nature of this hypothetical progression remains controversial. Antiepileptic drug treatment necessarily confounds analyses of progressive changes in human patients with acquired epilepsy. Here, we describe experiments testing the hypothesis that development of acquired epilepsy begins as a continuous process of increased seizure frequency (i.e., proportional to probability of a spontaneous seizure) that ultimately plateaus. Using nearly continuous surface cortical and bilateral hippocampal recordings with radiotelemetry and semiautomated seizure detection, the frequency of electrographically recorded seizures (both convulsive and nonconvulsive) was analyzed quantitatively for approximately 100 d after kainate-induced status epilepticus in adult rats. The frequency of spontaneous recurrent seizures was not a step function of time (as implied by the "latent period"); rather, seizure frequency increased as a sigmoid function of time. The distribution of interseizure intervals was nonrandom, suggesting that seizure clusters (i.e., short interseizure intervals) obscured the early stages of progression, and may have contributed to the increase in seizure frequency. These data suggest that (1) the latent period is the first of many long interseizure intervals and a poor measure of the time frame of epileptogenesis, (2) epileptogenesis is a continuous process that extends much beyond the first spontaneous recurrent seizure, (3) uneven seizure clustering contributes to the variability in occurrence of epileptic seizures, and (4) the window for antiepileptogenic therapies aimed at suppressing acquired epilepsy probably extends well past the first clinical seizure.
Figures
Comment in
-
The window of epileptogenesis: looking beyond the latent period.Epilepsy Curr. 2009 Sep-Oct;9(5):144-5. doi: 10.1111/j.1535-7511.2009.01322.x. Epilepsy Curr. 2009. PMID: 19826508 Free PMC article. No abstract available.
References
-
- Annegers JF, Hauser WA, Coan SP, Rocca WA. A population-based study of seizures after traumatic brain injuries. N Engl J Med. 1998;338:20–24. - PubMed
-
- Ben-Ari Y. Limbic seizure and brain damage produced by kainic acid: mechanisms and relevance to human temporal lobe epilepsy. Neuroscience. 1985;14:375–403. - PubMed
-
- Bertram EH. Functional anatomy of spontaneous seizures in a rat model of limbic epilepsy. Epilpesia. 1997;38:95–105. - PubMed
-
- Bertram EH, Cornett JF. The ontogeny of seizures in a rat model of limbic epilepsy: evidence for a kindling process in the development of chronic spontaneous seizures. Brain Res. 1993;625:295–300. - PubMed
-
- Bertram EH, Cornett JF. The evolution of a rat model of chronic spontaneous limbic seizures. Brain Res. 1994;661:157–162. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
