Sphingolipid signaling and treatment during remodeling of the uninfarcted ventricular wall after myocardial infarction
- PMID: 19234089
- PMCID: PMC2670694
- DOI: 10.1152/ajpheart.01032.2008
Sphingolipid signaling and treatment during remodeling of the uninfarcted ventricular wall after myocardial infarction
Abstract
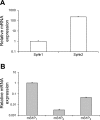
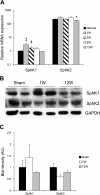
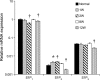
The sphingosine kinase (SphK)/sphingosine 1-phosphate (S1P) pathway, known to determine the fate and growth of various cell types, can enhance cardiac myocyte survival in vitro and provide cardioprotection in acute ex vivo heart preparations. However, the relevance of these findings to chronic cardiac pathology has never been demonstrated. We hypothesized that S1P signaling is impaired during chronic remodeling of the uninfarcted ventricle during the evolution of post-myocardial infarction (MI) cardiomyopathy and that a therapeutic enhancement of S1P signaling would ameliorate ventricular dysfunction. SphK expression and activity were measured in the remote, uninfarcted myocardium (RM) of C57Bl/6 mice subjected to coronary artery ligation. The mRNA expression of S1P receptor isoforms was also measured, as was the activation of the downstream S1P receptor mediators. A cardioprotective role for S1P(1) receptor agonism was tested via the administration of the S1P(1)-selective agonist SEW2871 during and after MI. As a result, the expression data suggested that a dramatic reduction in SphK activity in the RM early after MI may reflect a combination of posttranscriptional and posttranslational modulation. SphK activity continued to decline gradually during chronic post-MI remodeling, when S1P(1) receptor mRNA also fell below baseline. The S1P(1)-specific agonism with oral SEW2871 during the first 2-wk after MI reduced apoptosis in the RM and resulted in improved myocardial function, as reflected in the echocardiographic measurement of fractional shortening. In conclusion, these results provide the first documentation of alterations in S1P-mediated signaling during the in situ development of cardiomyopathy and suggest a possible therapeutic role for the pharmacological S1P receptor agonism in the post-MI heart.
Figures

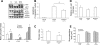
Similar articles
-
Sphingosine 1-phosphate signaling contributes to cardiac inflammation, dysfunction, and remodeling following myocardial infarction.Am J Physiol Heart Circ Physiol. 2016 Jan 15;310(2):H250-61. doi: 10.1152/ajpheart.00372.2015. Epub 2015 Nov 20. Am J Physiol Heart Circ Physiol. 2016. PMID: 26589326
-
Sphingosine-1-Phosphate Receptor Agonist Fingolimod Increases Myocardial Salvage and Decreases Adverse Postinfarction Left Ventricular Remodeling in a Porcine Model of Ischemia/Reperfusion.Circulation. 2016 Mar 8;133(10):954-66. doi: 10.1161/CIRCULATIONAHA.115.012427. Epub 2016 Jan 29. Circulation. 2016. PMID: 26826180
-
Sphingosine-1-phosphate interactions in the spleen and heart reflect extent of cardiac repair in mice and failing human hearts.Am J Physiol Heart Circ Physiol. 2021 Sep 1;321(3):H599-H611. doi: 10.1152/ajpheart.00314.2021. Epub 2021 Aug 20. Am J Physiol Heart Circ Physiol. 2021. PMID: 34415189 Free PMC article.
-
Expansion of Sphingosine Kinase and Sphingosine-1-Phosphate Receptor Function in Normal and Cancer Cells: From Membrane Restructuring to Mediation of Estrogen Signaling and Stem Cell Programming.Int J Mol Sci. 2018 Jan 31;19(2):420. doi: 10.3390/ijms19020420. Int J Mol Sci. 2018. PMID: 29385066 Free PMC article. Review.
-
New players on the center stage: sphingosine 1-phosphate and its receptors as drug targets.Biochem Pharmacol. 2008 May 15;75(10):1893-900. doi: 10.1016/j.bcp.2007.12.018. Epub 2008 Jan 5. Biochem Pharmacol. 2008. PMID: 18321471 Review.
Cited by
-
Sphingolipid De Novo Biosynthesis: A Rheostat of Cardiovascular Homeostasis.Trends Endocrinol Metab. 2016 Nov;27(11):807-819. doi: 10.1016/j.tem.2016.07.005. Epub 2016 Aug 22. Trends Endocrinol Metab. 2016. PMID: 27562337 Free PMC article. Review.
-
U-shaped association between plasma sphingosine-1-phosphate levels and mortality in patients with chronic systolic heart failure: a prospective cohort study.Lipids Health Dis. 2020 Jun 4;19(1):125. doi: 10.1186/s12944-020-01262-2. Lipids Health Dis. 2020. PMID: 32498720 Free PMC article.
-
Sphingosine 1-Phosphate Receptors: Do They Have a Therapeutic Potential in Cardiac Fibrosis?Front Pharmacol. 2017 Jun 2;8:296. doi: 10.3389/fphar.2017.00296. eCollection 2017. Front Pharmacol. 2017. PMID: 28626422 Free PMC article. Review.
-
Cardiomyocyte S1P1 receptor-mediated extracellular signal-related kinase signaling and desensitization.J Cardiovasc Pharmacol. 2009 Jun;53(6):486-94. doi: 10.1097/FJC.0b013e3181a7b58a. J Cardiovasc Pharmacol. 2009. PMID: 19433984 Free PMC article.
-
Editorial: Extracellular vesicles in cardiovascular inflammation and calcification.Front Cardiovasc Med. 2022 Nov 8;9:1077124. doi: 10.3389/fcvm.2022.1077124. eCollection 2022. Front Cardiovasc Med. 2022. PMID: 36426218 Free PMC article. No abstract available.
References
-
- Alvarez SE, Milstien S, Spiegel S. Autocrine and paracrine roles of sphingosine-1-phosphate. Trends Endocrinol Metab 18: 300–307, 2007. - PubMed
-
- Armstrong PW Defining myocardial infarction: a work in progress: ischemic heart disease. Heart 94: 1076–1079, 2008. - PubMed
-
- Betito S, Cuvillier C. Regulation by sphingosine 1-phosphate of Bax and Bad activities during apoptosis in a MEK-dependent manner. Biochem Biophys Res Commun 340: 1273–1277, 2006. - PubMed
-
- Billich A, Bornancin F, Dévay P, Mechtcheriakova D, Urtz N, Baumruker T. Phosphorylation of the immunomodulatory drug FTY720 by sphingosine kinases. J Biol Chem 278: 47408–47415, 2003. - PubMed
-
- Cuvillier O, Pirianov G, Kleuser B, Vanek PG, Coso OA, Gutkind S, Spiegel S. Suppression of ceramide-mediated programmed cell death by sphingosine-1-phosphate. Nature 381: 800–803, 1996. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

