Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses
- PMID: 19234466
- PMCID: PMC2692245
- DOI: 10.1038/nsmb.1566
Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses
Abstract
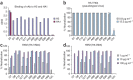
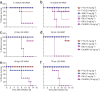
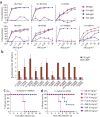
Influenza virus remains a serious health threat, owing to its ability to evade immune surveillance through rapid genetic drift and reassortment. Here we used a human non-immune antibody phage-display library and the H5 hemagglutinin ectodomain to select ten neutralizing antibodies (nAbs) that were effective against all group 1 influenza viruses tested, including H5N1 'bird flu' and the H1N1 'Spanish flu'. The crystal structure of one such nAb bound to H5 shows that it blocks infection by inserting its heavy chain into a conserved pocket in the stem region, thus preventing membrane fusion. Nine of the nAbs employ the germline gene VH1-69, and all seem to use the same neutralizing mechanism. Our data further suggest that this region is recalcitrant to neutralization escape and that nAb-based immunotherapy is a promising strategy for broad-spectrum protection against seasonal and pandemic influenza viruses.
Figures

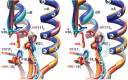
Comment in
-
Universal epitopes of influenza virus hemagglutinins?Nat Struct Mol Biol. 2009 Mar;16(3):233-4. doi: 10.1038/nsmb.1574. Epub 2009 Feb 22. Nat Struct Mol Biol. 2009. PMID: 19234464 No abstract available.
References
-
- WHO. Factsheet 211: influenza. World Health Organization 〈http://www.who.int/mediacentre/factsheets/2003/fs211/en/〉 (2003).
-
- WHO. Global influenza surveillance. World Health Organization 〈http://www.who.int/csr/disease/influenza/influenzanetwork/en/index.html〉 (2008).
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

