Glycine inhibitory dysfunction induces a selectively dynamic, morphine-resistant, and neurokinin 1 receptor- independent mechanical allodynia
- PMID: 19244526
- PMCID: PMC6666240
- DOI: 10.1523/JNEUROSCI.3923-08.2009
Glycine inhibitory dysfunction induces a selectively dynamic, morphine-resistant, and neurokinin 1 receptor- independent mechanical allodynia
Abstract
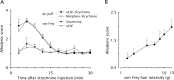
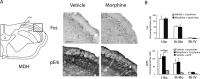
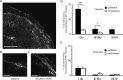
Dynamic mechanical allodynia is a widespread and intractable symptom of neuropathic pain for which there is a lack of effective therapy. We recently provided a novel perspective on the mechanisms of this symptom by showing that a simple switch in trigeminal glycine synaptic inhibition can turn touch into pain by unmasking innocuous input to superficial dorsal horn nociceptive specific neurons through a local excitatory, NMDA-dependent neural circuit involving neurons expressing the gamma isoform of protein kinase C. Here, we further investigated the clinical relevance and processing of glycine disinhibition. First, we showed that glycine disinhibition with strychnine selectively induced dynamic but not static mechanical allodynia. The induced allodynia was resistant to morphine. Second, morphine did not prevent the activation of the neural circuit underlying allodynia as shown by study of Fos expression and extracellular-signal regulated kinase phosphorylation in dorsal horn neurons. Third, in contrast to intradermal capsaicin injections, light, dynamic mechanical stimuli applied under disinhibition did not produce neurokinin 1 (NK1) receptor internalization in dorsal horn neurons. Finally, light, dynamic mechanical stimuli applied under disinhibition induced Fos expression only in neurons that did not express NK1 receptor. To summarize, the selectivity and morphine resistance of the glycine-disinhibition paradigm adequately reflect the clinical characteristics of dynamic mechanical allodynia. The present findings thus reveal the involvement of a selective dorsal horn circuit in dynamic mechanical allodynia, which operates through superficial lamina nociceptive-specific neurons that do not bear NK1 receptor and provide an explanation for the differences in the pharmacological sensitivity of neuropathic pain symptoms.
Figures



References
-
- Ahmadi S, Lippross S, Neuhuber WL, Zeilhofer HU. PGE(2) selectively blocks inhibitory glycinergic neurotransmission onto rat superficial dorsal horn neurons. Nat Neurosci. 2002;5:34–40. - PubMed
-
- Allen BJ, Li J, Menning PM, Rogers SD, Ghilardi J, Mantyh PW, Simone DA. Primary afferent fibers that contribute to increased substance P receptor internalization in the spinal cord after injury. J Neurophysiol. 1999;81:1379–1390. - PubMed
-
- Baccei M, Fitzgerald M. Development of pain pathways and mechanisms. In: McMahon SB, Koltzenburg M, editors. Wall and Melzack's textbook of pain. Oxford: Churchill Livingstone; 2006. pp. 143–158.
-
- Baron R. Mechanisms of disease: neuropathic pain–a clinical perspective. Nat Clin Pract Neurol. 2006;2:95–106. - PubMed
-
- Besse D, Lombard MC, Zajac JM, Roques BP, Besson JM. Pre- and postsynaptic distribution of mu, delta and kappa opioid receptors in the superficial layers of the cervical dorsal horn of the rat spinal cord. Brain Res. 1990;521:15–22. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
