Thiopeptide biosynthesis featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications
- PMID: 19246004
- PMCID: PMC2676563
- DOI: 10.1016/j.chembiol.2009.01.007
Thiopeptide biosynthesis featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications
Abstract
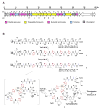
Thiopeptides, with potent activity against various drug-resistant pathogens, contain a characteristic macrocyclic core consisting of multiple thiazoles, dehydroamino acids, and a 6-membered nitrogen heterocycle. Their biosynthetic pathways remain elusive, in spite of great efforts by in vivo feeding experiments. Here, cloning, sequencing, and characterization of the thiostrepton and siomycin A gene clusters unveiled a biosynthetic paradigm for the thiopeptide specific core formation, featuring ribosomally synthesized precursor peptides and conserved posttranslational modifications. The paradigm generality for thiopeptide biosynthesis was supported by genome mining and ultimate confirmation of the thiocillin I production in Bacillus cereus ATCC 14579, a strain that was previously unknown as a thiopeptide producer. These findings set the stage to accelerate the discovery of thiopeptides by prediction at the genetic level and to generate structural diversity by applying combinatorial biosynthesis methods.
Figures
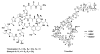

Similar articles
-
In vivo production of thiopeptide variants.Methods Enzymol. 2012;516:3-24. doi: 10.1016/B978-0-12-394291-3.00022-8. Methods Enzymol. 2012. PMID: 23034221
-
Nosiheptide biosynthesis featuring a unique indole side ring formation on the characteristic thiopeptide framework.ACS Chem Biol. 2009 Oct 16;4(10):855-64. doi: 10.1021/cb900133x. ACS Chem Biol. 2009. PMID: 19678698 Free PMC article.
-
Elucidating and engineering thiopeptide biosynthesis.World J Microbiol Biotechnol. 2017 Jun;33(6):119. doi: 10.1007/s11274-017-2283-9. Epub 2017 May 11. World J Microbiol Biotechnol. 2017. PMID: 28497389 Review.
-
ThioFinder: a web-based tool for the identification of thiopeptide gene clusters in DNA sequences.PLoS One. 2012;7(9):e45878. doi: 10.1371/journal.pone.0045878. Epub 2012 Sep 24. PLoS One. 2012. PMID: 23029291 Free PMC article.
-
Opportunities and challenges from current investigations into the biosynthetic logic of nosiheptide-represented thiopeptide antibiotics.Curr Opin Chem Biol. 2013 Aug;17(4):626-34. doi: 10.1016/j.cbpa.2013.06.021. Epub 2013 Jul 6. Curr Opin Chem Biol. 2013. PMID: 23838388 Review.
Cited by
-
The posttranslational modification cascade to the thiopeptide berninamycin generates linear forms and altered macrocyclic scaffolds.Proc Natl Acad Sci U S A. 2013 May 21;110(21):8483-8. doi: 10.1073/pnas.1307111110. Epub 2013 May 6. Proc Natl Acad Sci U S A. 2013. PMID: 23650400 Free PMC article.
-
Saturation mutagenesis of TsrA Ala4 unveils a highly mutable residue of thiostrepton A.ACS Chem Biol. 2015 Apr 17;10(4):998-1009. doi: 10.1021/cb5007745. Epub 2015 Jan 20. ACS Chem Biol. 2015. PMID: 25572285 Free PMC article.
-
Cerecidins, novel lantibiotics from Bacillus cereus with potent antimicrobial activity.Appl Environ Microbiol. 2014 Apr;80(8):2633-43. doi: 10.1128/AEM.03751-13. Epub 2014 Feb 14. Appl Environ Microbiol. 2014. PMID: 24532070 Free PMC article.
-
Thirtieth Anniversary of the Discovery of Laxaphycins. Intriguing Peptides Keeping a Part of Their Mystery.Mar Drugs. 2021 Aug 24;19(9):473. doi: 10.3390/md19090473. Mar Drugs. 2021. PMID: 34564135 Free PMC article. Review.
-
Antibiotics from microbes: converging to kill.Curr Opin Microbiol. 2009 Oct;12(5):520-7. doi: 10.1016/j.mib.2009.07.002. Epub 2009 Aug 18. Curr Opin Microbiol. 2009. PMID: 19695947 Free PMC article. Review.
References
-
- Bagley MC, Dale JW, Merritt EA, Xiong X. Thiopeptide antibiotics. Chem Rev. 2005;105:685–714. - PubMed
-
- Donia MS, Hathaway BJ, Sudek S, Haygood MG, Rosovitz MJ, Ravel J, Schmidt EW. Natural combinatorial peptide libraries in cyanobacterial symbionts of marine ascidians. Nat Chem Biol. 2006;2:729–735. - PubMed
-
- Donovick R, Pagano JF, Stout HA, Weinstein MJ. Thiostrepton, a new antibiotic. I In vitro studies. Antibiot Annu. 1955;3:554–559. - PubMed
-
- Dutcher JD, Vandeputte J. Thiostrepton, a new antibiotic. II Isolation and chemical characterization. Antibiot Annu. 1955;3:560–561. - PubMed
-
- Ebata M, Miyazaki K, Otsuka H. Studies on siomycin. I Physicochemical properties of siomycins A, B and C. J Antibiot (Tokyo) 1969;22:364–368. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

