Similar and distinct properties of MUPP1 and Patj, two homologous PDZ domain-containing tight-junction proteins
- PMID: 19255144
- PMCID: PMC2668367
- DOI: 10.1128/MCB.01505-08
Similar and distinct properties of MUPP1 and Patj, two homologous PDZ domain-containing tight-junction proteins
Abstract
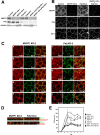
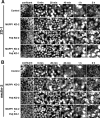
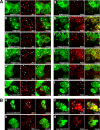
MUPP1 and Patj are both composed of an L27 domain and multiple PDZ domains (13 and 10 domains, respectively) and are localized to tight junctions (TJs) in epithelial cells. Although Patj is known to be responsible for the organization of TJs and epithelial polarity, characterization of MUPP1 is lacking. In this study, we found that MUPP1 and Patj share several binding partners, including JAM1, ZO-3, Pals1, Par6, and nectins (cell-cell adhesion molecules at adherens junctions). MUPP1 and Patj exhibited similar subcellular distributions, and the mechanisms with which they localize to TJs also appear to overlap. Despite these similarities, functional studies have revealed that Patj is indispensable for the establishment of TJs and epithelial polarization, whereas MUPP1 is not. Thus, although MUPP1 and Patj share several molecular properties, their functions are entirely different. We present evidence that the signaling mediated by Pals1, which has a higher affinity for Patj than for MUPP1 and is involved in the activation of the Par6-aPKC complex, is of principal importance for the function of Patj in epithelial cells.
Figures


Similar articles
-
The carboxyl terminus of zona occludens-3 binds and recruits a mammalian homologue of discs lost to tight junctions.J Biol Chem. 2002 Jul 26;277(30):27501-9. doi: 10.1074/jbc.M201177200. Epub 2002 May 20. J Biol Chem. 2002. PMID: 12021270
-
Molecular characterization of angiomotin/JEAP family proteins: interaction with MUPP1/Patj and their endogenous properties.Genes Cells. 2007 Apr;12(4):473-86. doi: 10.1111/j.1365-2443.2007.01066.x. Genes Cells. 2007. PMID: 17397395
-
The Maguk protein, Pals1, functions as an adapter, linking mammalian homologues of Crumbs and Discs Lost.J Cell Biol. 2002 Apr 1;157(1):161-72. doi: 10.1083/jcb.200109010. Epub 2002 Apr 1. J Cell Biol. 2002. PMID: 11927608 Free PMC article.
-
Epithelial junctions and Rho family GTPases: the zonular signalosome.Small GTPases. 2014;5(4):1-15. doi: 10.4161/21541248.2014.973760. Small GTPases. 2014. PMID: 25483301 Free PMC article. Review.
-
Structural and signalling molecules come together at tight junctions.Curr Opin Cell Biol. 1999 Oct;11(5):628-33. doi: 10.1016/s0955-0674(99)00016-2. Curr Opin Cell Biol. 1999. PMID: 10508648 Review.
Cited by
-
Novel Compound Heterozygous Variations in MPDZ Gene Caused Isolated Bilateral Macular Coloboma in a Chinese Family.Cells. 2022 Nov 14;11(22):3602. doi: 10.3390/cells11223602. Cells. 2022. PMID: 36429029 Free PMC article.
-
The circumferential actomyosin belt in epithelial cells is regulated by the Lulu2-p114RhoGEF system.Small GTPases. 2012 Apr-Jun;3(2):91-6. doi: 10.4161/sgtp.19112. Small GTPases. 2012. PMID: 22790195 Free PMC article.
-
Establishment of epithelial polarity--GEF who's minding the GAP?J Cell Sci. 2014 Aug 1;127(Pt 15):3205-15. doi: 10.1242/jcs.153197. Epub 2014 Jul 2. J Cell Sci. 2014. PMID: 24994932 Free PMC article. Review.
-
Tight junctions: from simple barriers to multifunctional molecular gates.Nat Rev Mol Cell Biol. 2016 Sep;17(9):564-80. doi: 10.1038/nrm.2016.80. Epub 2016 Jun 29. Nat Rev Mol Cell Biol. 2016. PMID: 27353478 Review.
-
Membrane prewetting by condensates promotes tight-junction belt formation.Nature. 2024 Aug;632(8025):647-655. doi: 10.1038/s41586-024-07726-0. Epub 2024 Aug 7. Nature. 2024. PMID: 39112699 Free PMC article.
References
-
- Ackermann, F., N. Zitranski, D. Heydecke, B. Wilhelm, T. Gudermann, and I. Boekhoff. 2008. The Multi-PDZ domain protein MUPP1 as a lipid raft-associated scaffolding protein controlling the acrosome reaction in mammalian spermatozoa. J. Cell. Physiol. 214757-768. - PubMed
-
- Anderson, J. M., and C. M. Van Itallie. 1995. Tight junctions and the molecular basis for regulation of paracellular permeability. Am. J. Physiol. 269G467-G475. - PubMed
-
- Ando-Akatsuka, Y., S. Yonemura, M. Itoh, M. Furuse, and S. Tsukita. 1999. Differential behavior of E-cadherin and occludin in their colocalization with ZO-1 during the establishment of epithelial cell polarity. J. Cell. Physiol. 179115-125. - PubMed
-
- Aoki, J., S. Koike, H. Asou, I. Ise, H. Suwa, T. Tanaka, M. Miyasaka, and A. Nomoto. 1997. Mouse homolog of poliovirus receptor-related gene 2 product, mPRR2, mediates homophilic cell aggregation. Exp. Cell Res. 235374-384. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
