Bcl-x L increases mitochondrial fission, fusion, and biomass in neurons
- PMID: 19255249
- PMCID: PMC2686401
- DOI: 10.1083/jcb.200809060
Bcl-x L increases mitochondrial fission, fusion, and biomass in neurons
Abstract
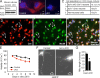
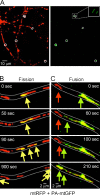
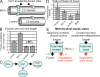
Mitochondrial fission and fusion are linked to synaptic activity in healthy neurons and are implicated in the regulation of apoptotic cell death in many cell types. We developed fluorescence microscopy and computational strategies to directly measure mitochondrial fission and fusion frequencies and their effects on mitochondrial morphology in cultured neurons. We found that the rate of fission exceeds the rate of fusion in healthy neuronal processes, and, therefore, the fission/fusion ratio alone is insufficient to explain mitochondrial morphology at steady state. This imbalance between fission and fusion is compensated by growth of mitochondrial organelles. Bcl-x(L) increases the rates of both fusion and fission, but more important for explaining the longer organelle morphology induced by Bcl-x(L) is its ability to increase mitochondrial biomass. Deficits in these Bcl-x(L)-dependent mechanisms may be critical in neuronal dysfunction during the earliest phases of neurodegeneration, long before commitment to cell death.
Figures


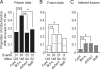

References
-
- Benard G., Rossignol R. 2008. Ultrastructure of the mitochondrion and its bearing on function and bioenergetics.Antioxid. Redox Signal. 10:1313–1342 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

