Influence of Ecto-nucleoside triphosphate diphosphohydrolase activity on Trypanosoma cruzi infectivity and virulence
- PMID: 19255624
- PMCID: PMC2644763
- DOI: 10.1371/journal.pntd.0000387
Influence of Ecto-nucleoside triphosphate diphosphohydrolase activity on Trypanosoma cruzi infectivity and virulence
Abstract
Background: The protozoan Trypanosoma cruzi is the causative agent of Chagas disease. There are no vaccines or effective treatment, especially in the chronic phase when most patients are diagnosed. There is a clear necessity to develop new drugs and strategies for the control and treatment of Chagas disease. Recent papers have suggested the ecto-nucleotidases (from CD39 family) from pathogenic agents as important virulence factors. In this study we evaluated the influence of Ecto-Nucleoside-Triphosphate-Diphosphohydrolase (Ecto-NTPDase) activity on infectivity and virulence of T. cruzi using both in vivo and in vitro models.
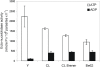
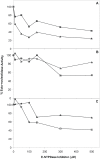
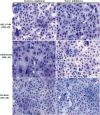
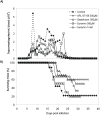
Methodology/principal findings: We followed Ecto-NTPDase activities of Y strain infective forms (trypomastigotes) obtained during sequential sub-cultivation in mammalian cells. ATPase/ADPase activity ratios of cell-derived trypomastigotes decreased 3- to 6-fold and infectivity was substantially reduced during sequential sub-cultivation. Surprisingly, at third to fourth passages most of the cell-derived trypomastigotes could not penetrate mammalian cells and had differentiated into amastigote-like parasites that exhibited 3- to 4-fold lower levels of Ecto-NTPDase activities. To evidence the participation of T. cruzi Ecto-NTPDase1 in the infective process, we evaluated the effect of known Ecto-ATPDase inhibitors (ARL 67156, Gadolinium and Suramin), or anti-NTPDase-1 polyclonal antiserum on ATPase and ADPase hydrolytic activities in recombinant T. cruzi NTPDase-1 and in live trypomastigotes. All tests showed a partial inhibition of Ecto-ATPDase activities and a marked inhibition of trypomastigotes infectivity. Mice infections with Ecto-NTPDase-inhibited trypomastigotes produced lower levels of parasitemia and higher host survival than with non-inhibited control parasites.
Conclusions/significance: Our results suggest that Ecto-ATPDases act as facilitators of infection and virulence in vitro and in vivo and emerge as target candidates in chemotherapy of Chagas disease.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


Similar articles
-
Ectonucleotidases from trypomastigotes from different sources and various genetic backgrounds of Trypanosoma cruzi potentiate their infectivity and host inflammation.Cytokine. 2020 Dec;136:155255. doi: 10.1016/j.cyto.2020.155255. Epub 2020 Aug 28. Cytokine. 2020. PMID: 32866897
-
Protective immunity triggered by ectonucleoside triphosphate diphosphohydrolase-based biopharmaceuticals attenuates cardiac parasitism and prevents mortality in Trypanosoma cruzi infection.Bioorg Med Chem. 2022 Oct 15;72:116966. doi: 10.1016/j.bmc.2022.116966. Epub 2022 Aug 17. Bioorg Med Chem. 2022. PMID: 35998390
-
A bacterial ecto-triphosphate diphosphohydrolase similar to human CD39 is essential for intracellular multiplication of Legionella pneumophila.Cell Microbiol. 2007 Aug;9(8):1922-35. doi: 10.1111/j.1462-5822.2007.00924.x. Epub 2007 Mar 26. Cell Microbiol. 2007. PMID: 17388784
-
NTPDase activities: possible roles on Leishmania spp infectivity and virulence.Cell Biol Int. 2018 Jun;42(6):670-682. doi: 10.1002/cbin.10944. Epub 2018 Feb 19. Cell Biol Int. 2018. PMID: 29384228 Review.
-
ATP-Diphosphohydrolases in Parasites: Localization, Functions and Recent Developments in Drug Discovery.Curr Protein Pept Sci. 2019;20(9):873-884. doi: 10.2174/1389203720666190704152827. Curr Protein Pept Sci. 2019. PMID: 31272352 Review.
Cited by
-
Leishmania infantum ecto-nucleoside triphosphate diphosphohydrolase-2 is an apyrase involved in macrophage infection and expressed in infected dogs.PLoS Negl Trop Dis. 2014 Nov 13;8(11):e3309. doi: 10.1371/journal.pntd.0003309. eCollection 2014 Nov. PLoS Negl Trop Dis. 2014. PMID: 25393008 Free PMC article.
-
Ecto-nucleotidase activities of promastigotes from Leishmania (Viannia) braziliensis relates to parasite infectivity and disease clinical outcome.PLoS Negl Trop Dis. 2012;6(10):e1850. doi: 10.1371/journal.pntd.0001850. Epub 2012 Oct 11. PLoS Negl Trop Dis. 2012. PMID: 23071853 Free PMC article.
-
Synthesis of new non-natural L-glycosidic flavonoid derivatives and their evaluation as inhibitors of Trypanosoma cruzi ecto-nucleoside triphosphate diphosphohydrolase 1 (TcNTPDase1).Purinergic Signal. 2024 Aug;20(4):399-419. doi: 10.1007/s11302-023-09974-7. Epub 2023 Nov 17. Purinergic Signal. 2024. PMID: 37975950 Free PMC article.
-
ATPe Dynamics in Protozoan Parasites. Adapt or Perish.Genes (Basel). 2018 Dec 27;10(1):16. doi: 10.3390/genes10010016. Genes (Basel). 2018. PMID: 30591699 Free PMC article. Review.
-
Involvement of ectonucleotidases and purinergic receptor expression during acute Chagas disease in the cortex of mice treated with resveratrol and benznidazole.Purinergic Signal. 2021 Sep;17(3):493-502. doi: 10.1007/s11302-021-09803-9. Epub 2021 Jul 24. Purinergic Signal. 2021. PMID: 34302569 Free PMC article.
References
-
- TDR. Progress 2003–2004, Tropical Disease Research, Making Health Research Work for Poor People. 2005. TDR/GEN/05.1.
-
- Guedes PMM, Fietto JLR, Lana M, Bahia MT. Advances in Chagas Disease Chemotherapy. Anti-Infective Agents in Med Chem. 2006;5:175–186.
-
- Matsuoka I, Ohkubo S. ATP- and adenosine-mediated signaling in the central nervous system: adenosine receptor activation by ATP through rapid and localized generation of adenosine by ecto-nucleotidases. J Pharmacol Sci. 2004;94:95–99. - PubMed
-
- Di Virgilio F, Chiozzi P, Ferrari D, Falzoni S, Sanz JM, et al. Nucleotide receptors: an emerging family of regulatory molecules in blood cells. Blood. 2001;97:587–600. - PubMed
-
- Mizumoto N, Kumamoto T, Robson SC, Sevigny J, Matsue H, et al. CD39 is the dominant Langerhans cell-associated ecto-NTPDase: modulatory roles in inflammation and immune responsiveness. Nat Med. 2002;8:358–365. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

