PML depletion disrupts normal mammary gland development and skews the composition of the mammary luminal cell progenitor pool
- PMID: 19261859
- PMCID: PMC2660741
- DOI: 10.1073/pnas.0807640106
PML depletion disrupts normal mammary gland development and skews the composition of the mammary luminal cell progenitor pool
Abstract
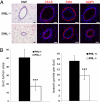
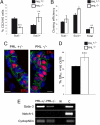
Nuclear domains of promyelocytic leukemia protein (PML) are known to act as signaling nodes in many cellular processes. Although the impact of PML expression in driving cell fate decisions for injured cells is well established, the function of PML in the context of tissue development is less well understood. Here, the in vivo role of PML in developmental processes in the murine mammary gland has been investigated. Data are presented showing that expression of PML is tightly regulated by three members of the Stat family of transcription factors that orchestrate the functional development of the mammary secretory epithelium during pregnancy. Developmental phenotypes were also discovered in the virgin and pregnant Pml null mouse, typified by aberrant differentiation of mammary epithelia with reduced ductal and alveolar development. PML depletion was also found to disturb the balance of two distinct luminal progenitor populations. Overall, it is shown that PML is required for cell lineage determination in bi-potent luminal progenitor cells and that the precise regulation of PML expression is required for functional differentiation of alveolar cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Salomoni P, Ferguson BJ, Wyllie AH, Rich T. New insights into the role of PML in tumour suppression. Cell Res. 2008;18:622–640. - PubMed
-
- Bernardi R, Pandolfi PP. Structure, dynamics and functions of promyelocytic leukaemia nuclear bodies. Nat Rev Mol Cell Biol. 2007;8:1006–1016. - PubMed
-
- Melnick A, Licht JD. Deconstructing a disease: RARalpha, its fusion partners, and their roles in the pathogenesis of acute promyelocytic leukemia. Blood. 1999;93:3167–3215. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

