Heat shock protein gp96 interacts with protein phosphatase 5 and controls toll-like receptor 2 (TLR2)-mediated activation of extracellular signal-regulated kinase (ERK) 1/2 in post-hypoxic kidney cells
- PMID: 19265198
- PMCID: PMC2673320
- DOI: 10.1074/jbc.M808376200
Heat shock protein gp96 interacts with protein phosphatase 5 and controls toll-like receptor 2 (TLR2)-mediated activation of extracellular signal-regulated kinase (ERK) 1/2 in post-hypoxic kidney cells
Abstract
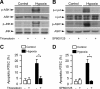
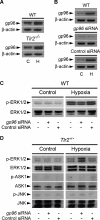
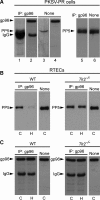
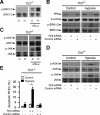
Ischemia/reperfusion injury (IRI) induces an innate immune response, leading to an inflammatory reaction and tissue damage that have been attributed to engagement of the Toll-like receptor (TLR) 2 and 4. However, the respective roles of TLR2 and/or TLR4 in mediating downstream activation of mitogen-activated protein kinase (MAPK) pathways during IRI have not been fully elucidated. Here we show that extracellular signal-regulated kinase (ERK)1/2 is activated in both intact kidneys and cultured renal tubule epithelial cells (RTECs) from wildtype and Tlr4 knockout mice, but not those from Tlr2 knockout mice subjected to transient ischemia. Geldanamycin (GA), an inhibitor of heat shock protein 90 and reticulum endoplasmic-resident gp96, and gp96 mRNA silencing (siRNA), did not affect ERK1/2 activation in either post-hypoxic wild-type or Tlr4-deficient RTECs, but did restore its activation in post-hypoxic Tlr2-deficient RTECs. Immunoprecipitation studies revealed that gp96 co-immunoprecipitates with the serine-threonine protein phosphatase 5 (PP5), identified as a negative modulator of the mitogen extracellular kinase (MEK)-ERK pathway, in unstressed wild-type and post-hypoxic Tlr2-deficient RTECs. In contrast, PP5 co-immunoprecipitation with gp96 was strikingly reduced in post-hypoxic wild-type RTECs, suggesting that the inactivation of PP5 resulting from the dissociation of PP5 from gp96 allows the activation of ERK1/2 to occur. Inhibition of PP5 by okadaic acid, and Pp5 siRNA also restored TLR2-mediated phosphorylation of ERK1/2, and apoptosis signal-regulating kinase 1 (ASK1)/c-Jun N-terminal kinase (JNK)-mediated apoptosis in post-hypoxic Tlr2-deficient RTECs. These findings indicate that gp96 interacts with PP5 and controls TLR2-mediated induction of ERK1/2 in post-hypoxic renal tubule cells.
Figures



References
-
- Bonventre, J. V., and Weinberg, J. M. (2003) J. Am. Soc. Nephrol. 14 2199-2210 - PubMed
-
- Beg, A. A. (2002) Trends Immunol. (2002) 23 509-512 - PubMed
-
- Akira, S., and Takeda, K. (2004) Nat. Rev. Immunol. 4 499-511 - PubMed
-
- Tsuboi, N., Yoshikai, Y., Matsuo, S., Kikuchi, T., Iwami, K., Nagai, Y., Takeuchi, O., Akira, S., and Matsuguchi, T. (2002) J. Immunol. 169 2026-2033 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

