Quantification of intermediates formed during the reduction of nitrite by deoxyhemoglobin
- PMID: 19270306
- PMCID: PMC2676000
- DOI: 10.1074/jbc.M808647200
Quantification of intermediates formed during the reduction of nitrite by deoxyhemoglobin
Abstract
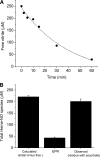
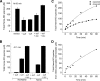
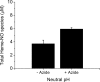
Nitric oxide (NO) plays a crucial role in human physiology by regulating vascular tone and blood flow. The short life-span of NO in blood requires a mechanism to retain NO bioactivity in the circulation. Recent studies have suggested a mechanism involving the reduction of nitrite back to NO by deoxyhemoglobin in RBCs. A role for RBCs in transporting NO must, however, bypass the scavenging of NO in RBCs by hemoglobin. To understand how the nitrite reaction can deliver bioactive NO to the vasculature, we have studied the intermediates formed during the reaction. A reliable measure of the total concentration of heme-associated nitrite/NO intermediates formed was provided by combining filtration to measure free nitrite by chemiluminescence and electron paramagnetic resonance to measure the final product Hb(II)NO. By modifying the chemiluminescence method used to detect NO, we have been able to identify two intermediates: 1) a heme-associated nitrite complex that is released as NO in acid solution in the presence of ascorbate and 2) an intermediate that releases NO at neutral pH in the presence of ferricyanide when reacted with an Fe(III) ligand like azide. This species designated as "Hb(II)NO(+) (<--)(-->)Hb(III)NO" has properties of both isomeric forms resulting in a slower NO dissociation rate and much higher stability than Hb(III)NO, but provides a potential source for bioactive NO, which can be released from the RBC. This detailed analysis of the nitrite reaction with deoxyHb provides important insights into the mechanism for nitrite induced vasodilation by RBCs.
Figures


Similar articles
-
Red blood cell membrane-facilitated release of nitrite-derived nitric oxide bioactivity.Biochemistry. 2015 Nov 10;54(44):6712-23. doi: 10.1021/acs.biochem.5b00643. Epub 2015 Oct 28. Biochemistry. 2015. PMID: 26478948
-
A new paramagnetic intermediate formed during the reaction of nitrite with deoxyhemoglobin.J Am Chem Soc. 2011 Aug 24;133(33):13010-22. doi: 10.1021/ja1115088. Epub 2011 Aug 2. J Am Chem Soc. 2011. PMID: 21755997 Free PMC article.
-
Intermediates detected by visible spectroscopy during the reaction of nitrite with deoxyhemoglobin: the effect of nitrite concentration and diphosphoglycerate.Biochemistry. 2007 Oct 16;46(41):11650-9. doi: 10.1021/bi700364e. Epub 2007 Sep 20. Biochemistry. 2007. PMID: 17880185
-
Nitric oxide redox reactions and red cell biology.Antioxid Redox Signal. 2006 Jul-Aug;8(7-8):1193-203. doi: 10.1089/ars.2006.8.1193. Antioxid Redox Signal. 2006. PMID: 16910767 Review.
-
The new chemical biology of nitrite reactions with hemoglobin: R-state catalysis, oxidative denitrosylation, and nitrite reductase/anhydrase.Acc Chem Res. 2009 Jan 20;42(1):157-67. doi: 10.1021/ar800089j. Acc Chem Res. 2009. PMID: 18783254 Review.
Cited by
-
Small molecule signaling agents: the integrated chemistry and biochemistry of nitrogen oxides, oxides of carbon, dioxygen, hydrogen sulfide, and their derived species.Chem Res Toxicol. 2012 Apr 16;25(4):769-93. doi: 10.1021/tx2005234. Epub 2012 Feb 9. Chem Res Toxicol. 2012. PMID: 22263838 Free PMC article. Review.
-
Involvement of ferryl in the reaction between nitrite and the oxy forms of globins.J Biol Inorg Chem. 2014 Oct;19(7):1233-9. doi: 10.1007/s00775-014-1181-y. Epub 2014 Jul 27. J Biol Inorg Chem. 2014. PMID: 25064750
-
Nitrite enhances RBC hypoxic ATP synthesis and the release of ATP into the vasculature: a new mechanism for nitrite-induced vasodilation.Am J Physiol Heart Circ Physiol. 2009 Oct;297(4):H1494-503. doi: 10.1152/ajpheart.01233.2008. Epub 2009 Aug 21. Am J Physiol Heart Circ Physiol. 2009. PMID: 19700624 Free PMC article.
-
Potential Modulation of Vascular Function by Nitric Oxide and Reactive Oxygen Species Released From Erythrocytes.Front Physiol. 2018 Jun 7;9:690. doi: 10.3389/fphys.2018.00690. eCollection 2018. Front Physiol. 2018. PMID: 29930515 Free PMC article.
-
Extreme differences between hemoglobins I and II of the clam Lucina pectinalis in their reactions with nitrite.Biochim Biophys Acta. 2010 Oct;1804(10):1988-95. doi: 10.1016/j.bbapap.2010.06.016. Epub 2010 Jul 1. Biochim Biophys Acta. 2010. PMID: 20601225 Free PMC article.
References
-
- Ignarro, L. J., Cirino, G., Casini, A., and Napoli, C. (1999) J. Cardiovasc. Pharmacol. 34 879–886 - PubMed
-
- Gautier, C., van, F. E., Mikula, I., Martasek, P., and Slama-Schwok, A. (2006) Biochem. Biophys. Res. Commun. 341 816–821 - PubMed
-
- Liu, X., Miller, M. J., Joshi, M. S., Sadowska-Krowicka, H., Clark, D. A., and Lancaster, J. R., Jr. (1998) J. Biol. Chem. 273 18709–18713 - PubMed
-
- Cosby, K., Partovi, K. S., Crawford, J. H., Patel, R. P., Reiter, C. D., Martyr, S., Yang, B. K., Waclawiw, M. A., Zalos, G., Xu, X., Huang, K. T., Shields, H., Kim-Shapiro, D. B., Schechter, A. N., Cannon, R. O., III, and Gladwin, M. T. (2003) Nat. Med. 9 1498–1505 - PubMed
-
- Nagababu, E., Ramasamy, S., Abernethy, D. R., and Rifkind, J. M. (2003) J. Biol. Chem. 278 46349–46356 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

