Exogenous glucose-dependent insulinotropic polypeptide worsens post prandial hyperglycemia in type 2 diabetes
- PMID: 19276444
- PMCID: PMC2682676
- DOI: 10.2337/db08-0958
Exogenous glucose-dependent insulinotropic polypeptide worsens post prandial hyperglycemia in type 2 diabetes
Abstract
Objective: Glucose-dependent insulinotropic polypeptide (GIP), unlike glucagon-like peptide (GLP)-1, lacks glucose-lowering properties in patients with type 2 diabetes. We designed this study to elucidate the underlying pathophysiology.
Research design and methods: Twenty-two insulin-naïve subjects with type 2 diabetes were given either synthetic human GIP (20 ng x kg(-1) x min(-1)) or placebo (normal saline) over 180 min, starting with the first bite of a mixed meal (plus 1 g of acetaminophen) on two separate occasions. Frequent blood samples were obtained over 6 h to determine plasma GIP, GLP-1, glucose, insulin, glucagon, resistin, and acetaminophen levels.
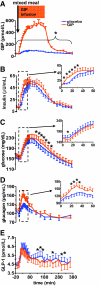
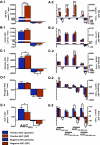
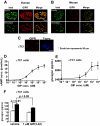
Results: Compared with placebo, GIP induced an early postprandial increase in insulin levels. Intriguingly, GIP also induced an early postprandial augmentation in glucagon, a significant elevation in late postprandial glucose, and a decrease in late postprandial GLP-1 levels. Resistin and acetaminophen levels were comparable in both interventions. By immunocytochemistry, GIP receptors were present on human and mouse alpha-cells. In alphaTC1 cell line, GIP induced an increase in intracellular cAMP and glucagon secretion. CONCLUSIONS; GIP, given to achieve supraphysiological plasma levels, still had an early, short-lived insulinotropic effect in type 2 diabetes. However, with a concomitant increase in glucagon, the glucose-lowering effect was lost. GIP infusion further worsened hyperglycemia postprandially, most likely through its suppressive effect on GLP-1. These findings make it unlikely that GIP or GIP receptor agonists will be useful in treating the hyperglycemia of patients with type 2 diabetes.
Figures

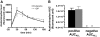
Similar articles
-
The pathophysiology of diabetes involves a defective amplification of the late-phase insulin response to glucose by glucose-dependent insulinotropic polypeptide-regardless of etiology and phenotype.J Clin Endocrinol Metab. 2003 Oct;88(10):4897-903. doi: 10.1210/jc.2003-030738. J Clin Endocrinol Metab. 2003. PMID: 14557471 Clinical Trial.
-
The insulinotropic actions of glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (7-37) in normal and diabetic subjects.Regul Pept. 1994 Apr 14;51(1):63-74. doi: 10.1016/0167-0115(94)90136-8. Regul Pept. 1994. PMID: 8036284
-
Xenin-25 delays gastric emptying and reduces postprandial glucose levels in humans with and without type 2 diabetes.Am J Physiol Gastrointest Liver Physiol. 2014 Feb 15;306(4):G301-9. doi: 10.1152/ajpgi.00383.2013. Epub 2013 Dec 19. Am J Physiol Gastrointest Liver Physiol. 2014. PMID: 24356886 Free PMC article. Clinical Trial.
-
Glucose-dependent insulinotropic polypeptide: effects on insulin and glucagon secretion in humans.Dan Med J. 2016 Apr;63(4):B5230. Dan Med J. 2016. PMID: 27034187 Review.
-
Potential role of tirzepatide towards Covid-19 infection in diabetic patients: a perspective approach.Inflammopharmacology. 2023 Aug;31(4):1683-1693. doi: 10.1007/s10787-023-01239-4. Epub 2023 May 19. Inflammopharmacology. 2023. PMID: 37208555 Free PMC article. Review.
Cited by
-
Glucagon's Metabolic Action in Health and Disease.Compr Physiol. 2021 Apr 1;11(2):1759-1783. doi: 10.1002/cphy.c200013. Compr Physiol. 2021. PMID: 33792899 Free PMC article.
-
Role of Gut-Related Peptides and Other Hormones in the Amelioration of Type 2 Diabetes after Roux-en-Y Gastric Bypass Surgery.ISRN Endocrinol. 2012;2012:504756. doi: 10.5402/2012/504756. Epub 2012 May 7. ISRN Endocrinol. 2012. PMID: 22619730 Free PMC article.
-
Enteroendocrine K and L cells in healthy and type 2 diabetic individuals.Diabetologia. 2018 Feb;61(2):284-294. doi: 10.1007/s00125-017-4450-9. Epub 2017 Sep 28. Diabetologia. 2018. PMID: 28956082
-
Current Therapies That Modify Glucagon Secretion: What Is the Therapeutic Effect of Such Modifications?Curr Diab Rep. 2017 Oct 28;17(12):128. doi: 10.1007/s11892-017-0967-z. Curr Diab Rep. 2017. PMID: 29080075 Review.
-
Incretin hormones and type 2 diabetes.Diabetologia. 2023 Oct;66(10):1780-1795. doi: 10.1007/s00125-023-05956-x. Epub 2023 Jul 11. Diabetologia. 2023. PMID: 37430117 Free PMC article. Review.
References
-
- Meier JJ, Nauck MA: Glucagon-like peptide 1(GLP-1) in biology and pathology. Diabetes Metab Res Rev 2005; 21: 91– 117 - PubMed
-
- Nauck MA, Heimesaat MM, Behle K, Holst JJ, Nauck MS, Ritzel R, Hufner M, Schmiegel WH: Effects of glucagon-like peptide 1 on counterregulatory hormone responses, cognitive functions, and insulin secretion during hyperinsulinemic, stepped hypoglycemic clamp experiments in healthy volunteers. J Clin Endocrinol Metab 2002; 87: 1239– 1246 - PubMed
-
- Nauck MA, Kleine N, Orskov C, Holst JJ, Willms B, Creutzfeldt W: Normalization of fasting hyperglycaemia by exogenous glucagon-like peptide 1 (7–36 amide) in type 2 (non-insulin-dependent) diabetic patients. Diabetologia 1993; 36: 741– 744 - PubMed
-
- Meier JJ, Gallwitz B, Salmen S, Goetze O, Holst JJ, Schmidt WE, Nauck MA: Normalization of glucose concentrations and deceleration of gastric emptying after solid meals during intravenous glucagon-like peptide 1 in patients with type 2 diabetes. J Clin Endocrinol Metab 2003; 88: 2719– 2725 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

