An essential role of the Forkhead-box transcription factor Foxo1 in control of T cell homeostasis and tolerance
- PMID: 19285438
- PMCID: PMC2692529
- DOI: 10.1016/j.immuni.2009.02.003
An essential role of the Forkhead-box transcription factor Foxo1 in control of T cell homeostasis and tolerance
Abstract
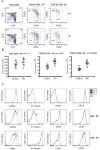
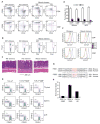
Members of the Forkhead box O (Foxo) family of transcription factors are key regulators of cellular responses, but their function in the immune system remains incompletely understood. Here we showed that T cell-specific deletion of Foxo1 gene in mice led to spontaneous T cell activation, effector T cell differentiation, autoantibody production, and the induction of inflammatory bowel disease in a transfer model. In addition, Foxo1 was critical for the maintenance of naive T cells in the peripheral lymphoid organs. Transcriptome analyses of T cells identified Foxo1-regulated genes encoding, among others, cell-surface molecules, signaling proteins, and nuclear factors that control gene expression. Functional studies validated interleukin-7 receptor-alpha as a Foxo1 target gene essential for Foxo1 maintenance of naive T cells. These findings reveal crucial functions of Foxo1-dependent transcription in control of T cell homeostasis and tolerance.
Conflict of interest statement
The authors declare that they have no competing financial interests.
Figures





References
-
- Akashi K, Kondo M, von Freeden-Jeffry U, Murray R, Weissman IL. Bcl-2 rescues T lymphopoiesis in interleukin-7 receptor-deficient mice. Cell. 1997;89:1033–1041. - PubMed
-
- Barata JT, Cardoso AA, Nadler LM, Boussiotis VA. Interleukin-7 promotes survival and cell cycle progression of T-cell acute lymphoblastic leukemia cells by down-regulating the cyclin-dependent kinase inhibitor p27(kip1) Blood. 2001;98:1524–1531. - PubMed
-
- Brunet A, Bonni A, Zigmond MJ, Lin MZ, Juo P, Hu LS, Anderson MJ, Arden KC, Blenis J, Greenberg ME. Akt promotes cell survival by phosphorylating and inhibiting a Forkhead transcription factor. Cell. 1999;96:857–868. - PubMed
-
- Burgering BM. A brief introduction to FOXOlogy. Oncogene. 2008;27:2258–2262. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

