The efficacy and safety of a new fixed-dose combination of amodiaquine and artesunate in young African children with acute uncomplicated Plasmodium falciparum
- PMID: 19291301
- PMCID: PMC2662869
- DOI: 10.1186/1475-2875-8-48
The efficacy and safety of a new fixed-dose combination of amodiaquine and artesunate in young African children with acute uncomplicated Plasmodium falciparum
Abstract
Background: Artesunate (AS) plus amodiaquine (AQ) is one artemisinin-based combination (ACT) recommended by the WHO for treating Plasmodium falciparum malaria. Fixed-dose AS/AQ is new, but its safety and efficacy are hitherto untested.
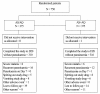
Methods: A randomized, open-label trial was conducted comparing the efficacy (non-inferiority design) and safety of fixed (F) dose AS (25 mg)/AQ (67.5 mg) to loose (L) AS (50 mg) + AQ (153 mg) in 750, P. falciparum-infected children from Burkina Faso aged 6 months to 5 years. Dosing was by age. Primary efficacy endpoint was Day (D) 28, PCR-corrected, parasitological cure rate. Recipients of rescue treatment were counted as failures and new infections as cured. Documented, common toxicity criteria (CTC) graded adverse events (AEs) defined safety.
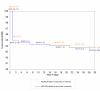
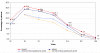
Results: Recruited and evaluable children numbered 750 (375/arm) and 682 (90.9%), respectively. There were 8 (AS/AQ) and 6 (AS+AQ) early treatment failures and one D7 failure (AS+AQ). Sixteen (AS/AQ) and 12 (AS+AQ) patients had recurrent parasitaemia (PCR new infections 10 and 6, respectively). Fourteen patients per arm required rescue treatment for vomiting/spitting out study drugs. Efficacy rates were 92.1% in both arms: AS/AQ = 315/342 (95% CI: 88.7-94.7) vs. AS+AQ = 313/340 (95% CI: 88.6-94.7). Non-inferiority was demonstrated at two-sided alpha = 0.05: Delta (AS+AQ - AS/AQ) = 0.0% (95% CI: -4.1% to 4.0%). D28, Kaplan Meier PCR-corrected cure rates (all randomized children) were similar: 93.7% (AS/AQ) vs. 93.2% (AS+AQ) Delta = -0.5 (95% CI -4.2 to 3.0%). By D2, both arms had rapid parasite (F & L, 97.8% aparasitaemic) and fever (97.2% [F], 96.0% [L] afebrile) clearances.Both treatments were well tolerated. Drug-induced vomiting numbered 8/375 (2.1%) and 6/375 (1.6%) in the fixed and loose arms, respectively (p = 0.59). One patient developed asymptomatic, CTC grade 4 hepatitis (AST 1052, ALT 936). Technical difficulties precluded the assessment and risk of neutropaenia for all patients.
Conclusion: Fixed dose AS/AQ was efficacious and well tolerated. These data support the use of this new fixed dose combination for treating P. falciparum malaria with continued safety monitoring.
Trial registration: Current Controlled Trials ISRCTN07576538.
Figures
Similar articles
-
Randomized, multicentre assessment of the efficacy and safety of ASAQ--a fixed-dose artesunate-amodiaquine combination therapy in the treatment of uncomplicated Plasmodium falciparum malaria.Malar J. 2009 Jun 8;8:125. doi: 10.1186/1475-2875-8-125. Malar J. 2009. PMID: 19505304 Free PMC article. Clinical Trial.
-
Artesunate-amodiaquine fixed dose combination for the treatment of Plasmodium falciparum malaria in India.Malar J. 2012 Mar 30;11:97. doi: 10.1186/1475-2875-11-97. Malar J. 2012. PMID: 22458860 Free PMC article. Clinical Trial.
-
In vivo/ex vivo efficacy of artemether-lumefantrine and artesunate-amodiaquine as first-line treatment for uncomplicated falciparum malaria in children: an open label randomized controlled trial in Burkina Faso.Malar J. 2020 Jan 6;19(1):8. doi: 10.1186/s12936-019-3089-z. Malar J. 2020. PMID: 31906948 Free PMC article. Clinical Trial.
-
Pyronaridine-artesunate for treating uncomplicated Plasmodium falciparum malaria.Cochrane Database Syst Rev. 2019 Jan 8;1(1):CD006404. doi: 10.1002/14651858.CD006404.pub3. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2022 Jun 21;6:CD006404. doi: 10.1002/14651858.CD006404.pub4. PMID: 30620055 Free PMC article. Updated.
-
Atovaquone-proguanil for treating uncomplicated Plasmodium falciparum malaria.Cochrane Database Syst Rev. 2021 Jan 15;1(1):CD004529. doi: 10.1002/14651858.CD004529.pub3. Cochrane Database Syst Rev. 2021. PMID: 33459345 Free PMC article.
Cited by
-
Plasmodium falciparum clearance in clinical studies of artesunate-amodiaquine and comparator treatments in sub-Saharan Africa, 1999-2009.Malar J. 2014 Mar 25;13:114. doi: 10.1186/1475-2875-13-114. Malar J. 2014. PMID: 24666562 Free PMC article.
-
Efficacy of artesunate-amodiaquine and artemether-lumefantrine fixed-dose combinations for the treatment of uncomplicated Plasmodium falciparum malaria among children aged six to 59 months in Nimba County, Liberia: an open-label randomized non-inferiority trial.Malar J. 2013 Jul 17;12:251. doi: 10.1186/1475-2875-12-251. Malar J. 2013. PMID: 23866774 Free PMC article. Clinical Trial.
-
Population pharmacokinetics of artesunate and amodiaquine in African children.Malar J. 2009 Aug 20;8:200. doi: 10.1186/1475-2875-8-200. Malar J. 2009. PMID: 19691851 Free PMC article. Clinical Trial.
-
Efficacy of artemether-lumefantrine and artesunate-amodiaquine as first line therapy of uncomplicated malaria in Burkina Faso, 11 years after policy change.Pan Afr Med J. 2020 Mar 10;35:68. doi: 10.11604/pamj.2020.35.68.20849. eCollection 2020. Pan Afr Med J. 2020. PMID: 32537072 Free PMC article. Clinical Trial.
-
Haemoglobin changes and risk of anaemia following treatment for uncomplicated falciparum malaria in sub-Saharan Africa.BMC Infect Dis. 2017 Jun 23;17(1):443. doi: 10.1186/s12879-017-2530-6. BMC Infect Dis. 2017. PMID: 28645255 Free PMC article.
References
-
- Antimalarial drug combination therapy: Report of a WHO Technical Consultation, 2001. World Health Organization; http://libdoc.who.int/hq/2001/WHO_CDS_RBM_2001.35.pdf
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

