Alcohol-induced protein hyperacetylation: mechanisms and consequences
- PMID: 19291822
- PMCID: PMC2658852
- DOI: 10.3748/wjg.15.1219
Alcohol-induced protein hyperacetylation: mechanisms and consequences
Abstract
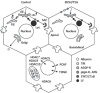
Although the clinical manifestations of alcoholic liver disease are well-described, little is known about the molecular basis of liver injury. Recent studies have indicated that ethanol exposure induces global protein hyperacetylation. This reversible, post-translational modification on the epsilon-amino groups of lysine residues has been shown to modulate multiple, diverse cellular processes ranging from transcriptional activation to microtubule stability. Thus, alcohol-induced protein hyperacetylation likely leads to major physiological consequences that contribute to alcohol-induced hepatotoxicity. Lysine acetylation is controlled by the activities of two opposing enzymes, histone acetyltransferases and histone deacetylases. Currently, efforts are aimed at determining which enzymes are responsible for the increased acetylation of specific substrates. However, the greater challenge will be to determine the physiological ramifications of protein hyperacetylation and how they might contribute to the progression of liver disease. In this review, we will first list and discuss the proteins known to be hyperacetylated in the presence of ethanol. We will then describe what is known about the mechanisms leading to increased protein acetylation and how hyperacetylation may perturb hepatic function.
Figures
References
-
- Brooks PJ. DNA damage, DNA repair, and alcohol toxicity--a review. Alcohol Clin Exp Res. 1997;21:1073–1082. - PubMed
-
- Kenney WC. Acetaldehyde adducts of phospholipids. Alcohol Clin Exp Res. 1982;6:412–416. - PubMed
-
- Kenney WC. Formation of Schiff base adduct between acetaldehyde and rat liver microsomal phosphatidylethanolamine. Alcohol Clin Exp Res. 1984;8:551–555. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

