Neural connectivity as an intermediate phenotype: brain networks under genetic control
- PMID: 19294651
- PMCID: PMC6871064
- DOI: 10.1002/hbm.20639
Neural connectivity as an intermediate phenotype: brain networks under genetic control
Abstract
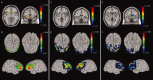
Recent evidence suggests that default mode connectivity characterizes neural states that account for a sizable proportion of brain activity and energy expenditure, and therefore represent a plausible neural intermediate phenotype. This implies the possibility of genetic control over systems-level connectivity features. Imaging genetics is an approach to combine genetic assessment with multimodal neuroimaging to discover neural systems linked to genetic abnormalities or variation. In the present contribution, we report results obtained from applying this strategy to both structural connectivity and functional connectivity data. Using data for serotonergic (5-HTTLPR, MAO-A) and dopaminergic (DARPP-32) genes as examples, we show that systems-level connectivity networks under genetic control can be identified. Remarkable similarities are observed across modalities and scales of description. Features of connectivity often better account for behavioral effects of genetic variation than regional parameters of activation or structure. These data provide convergent evidence for genetic control in humans over connectivity systems, whose characterization has promise for identifying neural systems mediating genetic risk for complex human behavior and psychiatric disease.
Copyright 2009 Wiley-Liss, Inc.
Figures


References
-
- Alexander GE,DeLong MR,Strick PL ( 1986): Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Annu Rev Neurosci 9: 357–381. - PubMed
-
- Buckholtz JW,Callicott JH,Kolachana B,Hariri AR,Goldberg TE,Genderson M,Egan MF,Mattay VS,Weinberger DR,Meyer‐Lindenberg A ( 2007a): Genetic variation in MAOA modulates ventromedial prefrontal circuitry mediating individual differences in human personality. Mol Psychiatry 13: 313–324. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

