Amyloid beta-induced neuronal hyperexcitability triggers progressive epilepsy
- PMID: 19295151
- PMCID: PMC6665248
- DOI: 10.1523/JNEUROSCI.5215-08.2009
Amyloid beta-induced neuronal hyperexcitability triggers progressive epilepsy
Abstract
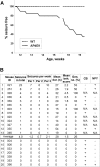
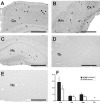
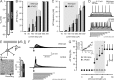
Alzheimer's disease is associated with an increased risk of unprovoked seizures. However, the underlying mechanisms of seizure induction remain elusive. Here, we performed video-EEG recordings in mice carrying mutant human APPswe and PS1dE9 genes (APdE9 mice) and their wild-type littermates to determine the prevalence of unprovoked seizures. In two recording episodes at the onset of amyloid beta (Abeta) pathogenesis (3 and 4.5 months of age), at least one unprovoked seizure was detected in 65% of APdE9 mice, of which 46% had multiple seizures and 38% had a generalized seizure. None of the wild-type mice had seizures. In a subset of APdE9 mice, seizure phenotype was associated with a loss of calbindin-D28k immunoreactivity in dentate granular cells and ectopic expression of neuropeptide Y in mossy fibers. In APdE9 mice, persistently decreased resting membrane potential in neocortical layer 2/3 pyramidal cells and dentate granule cells underpinned increased network excitability as identified by patch-clamp electrophysiology. At stimulus strengths evoking single-component EPSPs in wild-type littermates, APdE9 mice exhibited decreased action potential threshold and burst firing of pyramidal cells. Bath application (1 h) of Abeta1-42 or Abeta25-35 (proto-)fibrils but not oligomers induced significant membrane depolarization of pyramidal cells and increased the activity of excitatory cell populations as measured by extracellular field recordings in the juvenile rodent brain, confirming the pathogenic significance of bath-applied Abeta (proto-)fibrils. Overall, these data identify fibrillar Abeta as a pathogenic entity powerfully altering neuronal membrane properties such that hyperexcitability of pyramidal cells culminates in epileptiform activity.
Figures



Similar articles
-
Reduction of epileptiform activity by valproic acid in a mouse model of Alzheimer's disease is not long-lasting after treatment discontinuation.Epilepsy Res. 2015 May;112:43-55. doi: 10.1016/j.eplepsyres.2015.02.005. Epub 2015 Feb 14. Epilepsy Res. 2015. PMID: 25847338
-
Intrinsic excitability changes induced by acute treatment of hippocampal CA1 pyramidal neurons with exogenous amyloid β peptide.Hippocampus. 2015 Jul;25(7):786-97. doi: 10.1002/hipo.22403. Epub 2015 Mar 25. Hippocampus. 2015. PMID: 25515596 Free PMC article.
-
Suppression by topiramate of epileptiform burst discharges in hippocampal CA3 neurons of spontaneously epileptic rat in vitro.Brain Res. 1998 Apr 13;789(2):274-82. doi: 10.1016/s0006-8993(98)00116-4. Brain Res. 1998. PMID: 9573382
-
EPSPs of dentate gyrus granule cells during epileptiform bursts of dentate hilar "mossy" cells and area CA3 pyramidal cells in disinhibited rat hippocampal slices.J Neurosci. 1994 Oct;14(10):6041-57. doi: 10.1523/JNEUROSCI.14-10-06041.1994. J Neurosci. 1994. PMID: 7931561 Free PMC article.
-
Neuronal Hyperexcitability in APPSWE/PS1dE9 Mouse Models of Alzheimer's Disease.J Alzheimers Dis. 2021;81(3):855-869. doi: 10.3233/JAD-201540. J Alzheimers Dis. 2021. PMID: 33843674 Review.
Cited by
-
Major amyloid-β-degrading enzymes, endothelin-converting enzyme-2 and neprilysin, are expressed by distinct populations of GABAergic interneurons in hippocampus and neocortex.Neurobiol Aging. 2016 Dec;48:83-92. doi: 10.1016/j.neurobiolaging.2016.08.011. Epub 2016 Aug 20. Neurobiol Aging. 2016. PMID: 27644077 Free PMC article.
-
α-Synuclein forms non-selective cation channels and stimulates ATP-sensitive potassium channels in hippocampal neurons.J Physiol. 2015 Jan 1;593(1):145-59. doi: 10.1113/jphysiol.2014.280974. Epub 2014 Nov 13. J Physiol. 2015. PMID: 25556793 Free PMC article.
-
Intracerebral inoculation of healthy non-transgenic rats with a single aliquot of oligomeric amyloid-β (1-42) profoundly and progressively alters brain function throughout life.Front Aging Neurosci. 2024 Aug 2;16:1397901. doi: 10.3389/fnagi.2024.1397901. eCollection 2024. Front Aging Neurosci. 2024. PMID: 39156737 Free PMC article.
-
Spatial Memory Training Counteracts Hippocampal GIRK Channel Decrease in the Transgenic APPSw,Ind J9 Alzheimer's Disease Mouse Model.Int J Mol Sci. 2022 Nov 3;23(21):13444. doi: 10.3390/ijms232113444. Int J Mol Sci. 2022. PMID: 36362230 Free PMC article.
-
Linking activity dyshomeostasis and sleep disturbances in Alzheimer disease.Nat Rev Neurosci. 2024 Apr;25(4):272-284. doi: 10.1038/s41583-024-00797-y. Epub 2024 Feb 19. Nat Rev Neurosci. 2024. PMID: 38374463 Review.
References
-
- Amatniek JC, Hauser WA, DelCastillo-Castaneda C, Jacobs DM, Marder K, Bell K, Albert M, Brandt J, Stern Y. Incidence and predictors of seizures in patients with Alzheimer's disease. Epilepsia. 2006;47:867–872. - PubMed
-
- Breteler MM, Claus JJ, van Duijn CM, Launer LJ, Hofman A. Epidemiology of Alzheimer's disease. Epidemiol Rev. 1992;14:59–82. - PubMed
-
- Busche MA, Eichhoff G, Adelsberger H, Abramowski D, Wiederhold KH, Haass C, Staufenbiel M, Konnerth A, Garaschuk O. Clusters of hyperactive neurons near amyloid plaques in a mouse model of Alzheimer's disease. Science. 2008;321:1686–1689. - PubMed
-
- Carlson GA, Borchelt DR, Dake A, Turner S, Danielson V, Coffin JD, Eckman C, Meiners J, Nilsen SP, Younkin SG, Hsiao KK. Genetic modification of the phenotypes produced by amyloid precursor protein overexpression in transgenic mice. Hum Mol Genet. 1997;611:1951–1959. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
