Illuminating the chemistry of life: design, synthesis, and applications of "caged" and related photoresponsive compounds
- PMID: 19298086
- PMCID: PMC2700207
- DOI: 10.1021/cb900036s
Illuminating the chemistry of life: design, synthesis, and applications of "caged" and related photoresponsive compounds
Abstract
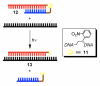
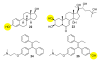
Biological systems are characterized by a level of spatial and temporal organization that often lies beyond the grasp of present day methods. Light-modulated bioreagents, including analogs of low molecular weight compounds, peptides, proteins, and nucleic acids, represent a compelling strategy to probe, perturb, or sample biological phenomena with the requisite control to address many of these organizational complexities. Although this technology has created considerable excitement in the chemical community, its application to biological questions has been relatively limited. We describe the challenges associated with the design, synthesis, and use of light-responsive bioreagents; the scope and limitations associated with the instrumentation required for their application; and recent chemical and biological advances in this field.
Figures
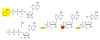


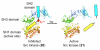



References
-
- Kornberg A. The two cultures: chemistry and biology. Biochemistry. 1987;26:6888–6891. - PubMed
-
- Kaplan JH, Forbush B, 3rd, Hoffman JF. Rapid photolytic release of adenosine 5′-triphosphate from a protected analogue: utilization by the Na:K pump of human red blood cell ghosts. Biochemistry. 1978;17:1929–1935. - PubMed
-
- Curley K, Lawrence DS. Caged regulators of signaling pathways. Pharmacol Ther. 1999;82:347–354. - PubMed
-
- Curley K, Lawrence DS. Light-activated proteins. Curr Opin Chem Biol. 1999;3:84–88. - PubMed
-
- Dorman G, Prestwich GD. Using photolabile ligands in drug discovery and development. Trends Biotechnol. 2000;18:64–77. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

