Trolox prevents osteoclastogenesis by suppressing RANKL expression and signaling
- PMID: 19299513
- PMCID: PMC2679474
- DOI: 10.1074/jbc.M806941200
Trolox prevents osteoclastogenesis by suppressing RANKL expression and signaling
Abstract
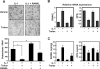
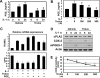
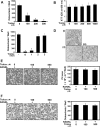
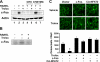
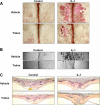
Excessive receptor activator of NF-kappaB ligand (RANKL) signaling causes enhanced osteoclast formation and bone resorption. Thus, down-regulation of RANKL expression or its downstream signals may be a therapeutic approach to the treatment of pathological bone loss. In this study, we investigated the effects of Trolox, a water-soluble vitamin E analogue, on osteoclastogenesis and RANKL signaling. Trolox potently inhibited interleukin-1-induced osteoclast formation in bone marrow cell-osteoblast coculture by abrogating RANKL induction in osteoblasts. This RANKL reduction was attributed to the reduced production of prostaglandin E(2) via a down-regulation of cyclooxygenase-2 activity. We also found that Trolox inhibited osteoclast formation from bone marrow macrophages induced by macrophage colony-stimulating factor plus RANKL in a reversible manner. Trolox was effective only when present during the early stage of culture, which implies that it targets early osteoclast precursors. Pretreatment with Trolox did not affect RANKL-induced early signaling pathways, including MAPKs, NF-kappaB, and Akt. We found that Trolox down-regulated the induction by RANKL of c-Fos protein by suppressing its translation. Ectopic overexpression of c-Fos rescued the inhibition of osteoclastogenesis by Trolox in bone marrow macrophages. Trolox also suppressed interleukin-1-induced osteoclast formation and bone loss in mouse calvarial bone. Taken together, our findings indicate that Trolox prevents osteoclast formation and bone loss by inhibiting both RANKL induction in osteoblasts and c-Fos expression in osteoclast precursors.
Figures


References
-
- Teitelbaum, S. L. (2000) Science 289 1504-1508 - PubMed
-
- Boyle, W. J., Simonet, W. S., and Lacey, D. L. (2003) Nature 423 337-342 - PubMed
-
- Suda, T., Nakamura, I., Jimi, E., and Takahashi, N. (1997) J. Bone Miner. Res. 12 869-879 - PubMed
-
- Lacey, D. L., Timms, E., Tan, H. L., Kelley, M. J., Dunstan, C. R., Burgess, T., Elliott, R., Colombero, A., Elliott, G., Scully, S., Hsu, H., Sullivan, J., Hawkins, N., Davy, E., Capparelli, C., Eli, A., Qian, Y. X., Kaufman, S., Sarosi, I., Shalhoub, V., Senaldi, G., Guo, J., Delaney, J., and Boyle, W. J. (1998) Cell 93 165-176 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

