The presence of a matrix-derived neutrophil chemoattractant in bronchiolitis obliterans syndrome after lung transplantation
- PMID: 19299743
- PMCID: PMC4219237
- DOI: 10.4049/jimmunol.0802457
The presence of a matrix-derived neutrophil chemoattractant in bronchiolitis obliterans syndrome after lung transplantation
Abstract
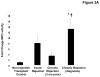
Lung transplantation is a therapeutic modality frequently used in end-stage lung disease. Unfortunately, lung transplant recipients have poor clinical outcomes, often due to the development of bronchiolitis obliterans syndrome (BOS). This process is often characterized by the pathologic findings of obliterative bronchiolitis: neutrophil influx and extracellular matrix remodeling leading to luminal obstruction and airway inflammation. The molecular mechanisms underlying BOS are poorly understood and disease-specific biomarkers are lacking. We report that in addition to increased levels of IL-8, the level of the neutrophil chemoattractant proline-glycine-proline (PGP) is elevated in BOS patient bronchoalveolar lavage (BAL) fluid. The enzymes responsible for generating PGP, matrix metalloproteases 8 and -9 and prolyl endopeptidase, are also elevated in these samples. Together, IL-8 and PGP account for most of the neutrophil chemoattractant capacity seen in BOS BAL fluid. Using specific neutralizing Abs to both IL-8 and PGP, we demonstrate that PGP is a prominent neutrophil chemoattractant found in BAL fluid from individuals at the time of diagnosis of BOS. These findings highlight the influence of a matrix-derived neutrophil chemoattractant in posttransplantation BOS and provide opportunities for the development of unique diagnostics and therapeutics to potentially improve disease outcomes.
Figures


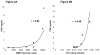
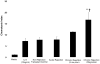

Similar articles
-
Airway neutrophilia in stable and bronchiolitis obliterans syndrome patients following lung transplantation.Thorax. 2000 Jan;55(1):53-9. doi: 10.1136/thorax.55.1.53. Thorax. 2000. PMID: 10607802 Free PMC article.
-
Broncho-alveolar lavage matrix metalloproteases as a sensitive measure of bronchiolitis obliterans.Am J Transplant. 2005 Jun;5(6):1548-52. doi: 10.1111/j.1600-6143.2005.00865.x. Am J Transplant. 2005. PMID: 15888067
-
A novel proteolytic cascade generates an extracellular matrix-derived chemoattractant in chronic neutrophilic inflammation.J Immunol. 2008 Apr 15;180(8):5662-9. doi: 10.4049/jimmunol.180.8.5662. J Immunol. 2008. PMID: 18390751 Free PMC article.
-
The role of neutrophils in the pathogenesis of obliterative bronchiolitis after lung transplantation.Transpl Infect Dis. 2001 Sep;3(3):168-76. doi: 10.1034/j.1399-3062.2001.003003168.x. Transpl Infect Dis. 2001. PMID: 11493399 Review.
-
Bronchoalveolar lavage as a tool to predict, diagnose and understand bronchiolitis obliterans syndrome.Am J Transplant. 2013 Mar;13(3):552-61. doi: 10.1111/ajt.12091. Epub 2013 Jan 28. Am J Transplant. 2013. PMID: 23356456 Free PMC article. Review.
Cited by
-
Sterile inflammation in thoracic transplantation.Cell Mol Life Sci. 2021 Jan;78(2):581-601. doi: 10.1007/s00018-020-03615-7. Epub 2020 Aug 17. Cell Mol Life Sci. 2021. PMID: 32803398 Free PMC article. Review.
-
Proline-Glycine-Proline (PGP) and High Mobility Group Box Protein-1 (HMGB1): Potential Mediators of Cystic Fibrosis Airway Inflammation.Open Respir Med J. 2010 Mar 30;4:32-8. doi: 10.2174/1874306401004020032. Open Respir Med J. 2010. PMID: 20448817 Free PMC article.
-
MMP-8 promotes polymorphonuclear cell migration through collagen barriers in obliterative bronchiolitis.J Leukoc Biol. 2010 Jan;87(1):69-77. doi: 10.1189/jlb.0509361. Epub 2009 Oct 2. J Leukoc Biol. 2010. PMID: 19801498 Free PMC article.
-
A CXCL8 receptor antagonist based on the structure of N-acetyl-proline-glycine-proline.Eur J Pharmacol. 2011 Oct 15;668(3):435-42. doi: 10.1016/j.ejphar.2011.02.045. Epub 2011 Mar 31. Eur J Pharmacol. 2011. PMID: 21458447 Free PMC article.
-
An extracellular matrix fragment drives epithelial remodeling and airway hyperresponsiveness.Sci Transl Med. 2018 Aug 22;10(455):eaaq0693. doi: 10.1126/scitranslmed.aaq0693. Sci Transl Med. 2018. PMID: 30135247 Free PMC article.
References
-
- Arcasoy SM, Kotloff RM. Lung transplantation. NEJM. 1999;340:1081–1089. - PubMed
-
- Trulock EP, Edwards LB, Taylor DO, Boucek MM, Keck BM, Hertz MI. Registry of the international society for heart and lung transplantation: Twenty-third official adult lung and heart-lung transplantation report 2006. The Journal of Heart and Lung Transplantation. 2006;25:880–892. - PubMed
-
- Estenne M, Maurer JR, Boehler A, Egan JJ, Frost A, Hertz MI, Mallory GB, Snell GI, Yousem S. Bronchiolitis obliterans syndrome 2001: An update of the diagnostic criteria. The Journal of Heart and Lung Transplantation. 2002;21:297–310. - PubMed
-
- Estenne M, Hertz MI. Bronchiolitis obliterans after human lung transplantation. Am J Respir Crit Care Med. 2002;166:440–444. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- S10 RR019231/RR/NCRR NIH HHS/United States
- HL090999/HL/NHLBI NIH HHS/United States
- T32A107493/PHS HHS/United States
- P30 DK074038/DK/NIDDK NIH HHS/United States
- HL07783/HL/NHLBI NIH HHS/United States
- P30 CA013148/CA/NCI NIH HHS/United States
- R01 HL102371/HL/NHLBI NIH HHS/United States
- P50 AT000477/AT/NCCIH NIH HHS/United States
- P30 CA13148/CA/NCI NIH HHS/United States
- M01 RR000032/RR/NCRR NIH HHS/United States
- R01 HL090999/HL/NHLBI NIH HHS/United States
- R01 HL077783/HL/NHLBI NIH HHS/United States
- U54 CA100949/CA/NCI NIH HHS/United States
- P30 AR050948/AR/NIAMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

