Compound heterozygous c-Mpl mutations in a child with congenital amegakaryocytic thrombocytopenia: functional characterization and a review of the literature
- PMID: 19302922
- PMCID: PMC2694734
- DOI: 10.1016/j.exphem.2009.01.001
Compound heterozygous c-Mpl mutations in a child with congenital amegakaryocytic thrombocytopenia: functional characterization and a review of the literature
Abstract
Objective: To genetically and functionally characterize mutations of c-Mpl that lead to thrombocytopenia in a child with congenital amegakaryocytic thrombocytopenia.
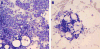
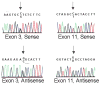
Materials and methods: We identified two c-Mpl mutations in a child with clinical features of congenital amegakaryocytic thrombocytopenia, one a previously described mutation in the extracellular domain (R102P) and the other a novel mutation leading to truncation of the receptor after the box 1 homology domain (541Stop). Cell line models were created to examine the ability of the mutant receptors to signal in response to thrombopoietin and thrombopoietin-like agonists.
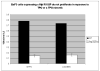
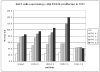
Results: Data from cell-line models indicate that c-Mpl R102P does not support significant signaling in response to thrombopoietin due to impaired trafficking of the mutant receptor to the cell surface. Alternative thrombopoietic agents do not circumvent this block to signaling, likely due to the inaccessibility of the receptor. In addition, previous data indicate that c-Mpl 541Stop does not support intracellular signaling due to the loss of critical intracellular domains.
Conclusions: This case demonstrates two different mechanisms by which c-Mpl mutations can impair thrombopoietin signaling, and suggests that mutations in the extracellular domain will not be rescued by c-Mpl agonists if they interfere with normal receptor expression.
Figures



References
-
- Ballmaier M, Germeshausen M, Schulze H, Cherkaoui K, Lang S, Gaudig A, Krukemeier S, Eilers M, Strauss G, Welte K. c-mpl mutations are the cause of congenital amegakaryocytic thrombocytopenia. Blood. 2001;97:139–46. - PubMed
-
- King S, Germeshausen M, Strauss G, Welte K, Ballmaier M. Congenital amegakaryocytic thrombocytopenia: a retrospective clinical analysis of 20 patients. Br J Haematol. 2005;131:636–44. - PubMed
-
- Onishi M, Mui AL, Morikawa Y, Cho L, Kinoshita S, Nolan GP, Gorman DM, Miyajima A, Kitamura T. Identification of an oncogenic form of the thrombopoietin receptor MPL using retrovirus-mediated gene transfer. Blood. 1996;88:1399–406. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

