Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial
- PMID: 19303137
- PMCID: PMC2915459
- DOI: 10.1016/S0140-6736(09)60500-6
Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial
Erratum in
- Lancet. 2009 Aug 8;374(9688):450
Abstract
Background: Gastrointestinal stromal tumour is the most common sarcoma of the intestinal tract. Imatinib mesylate is a small molecule that inhibits activation of the KIT and platelet-derived growth factor receptor alpha proteins, and is effective in first-line treatment of metastatic gastrointestinal stromal tumour. We postulated that adjuvant treatment with imatinib would improve recurrence-free survival compared with placebo after resection of localised, primary gastrointestinal stromal tumour.
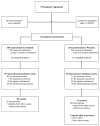
Methods: We undertook a randomised phase III, double-blind, placebo-controlled, multicentre trial. Eligible patients had complete gross resection of a primary gastrointestinal stromal tumour at least 3 cm in size and positive for the KIT protein by immunohistochemistry. Patients were randomly assigned, by a stratified biased coin design, to imatinib 400 mg (n=359) or to placebo (n=354) daily for 1 year after surgical resection. Patients and investigators were blinded to the treatment group. Patients assigned to placebo were eligible to crossover to imatinib treatment in the event of tumour recurrence. The primary endpoint was recurrence-free survival, and analysis was by intention to treat. Accrual was stopped early because the trial results crossed the interim analysis efficacy boundary for recurrence-free survival. This study is registered with ClinicalTrials.gov, number NCT00041197.
Findings: All randomised patients were included in the analysis. At median follow-up of 19.7 months (minimum-maximum 0-56.4), 30 (8%) patients in the imatinib group and 70 (20%) in the placebo group had had tumour recurrence or had died. Imatinib significantly improved recurrence-free survival compared with placebo (98% [95% CI 96-100] vs 83% [78-88] at 1 year; hazard ratio [HR] 0.35 [0.22-0.53]; one-sided p<0.0001). Adjuvant imatinib was well tolerated, with the most common serious events being dermatitis (11 [3%] vs 0), abdominal pain (12 [3%] vs six [1%]), and diarrhoea (ten [2%] vs five [1%]) in the imatinib group and hyperglycaemia (two [<1%] vs seven [2%]) in the placebo group.
Interpretation: Adjuvant imatinib therapy is safe and seems to improve recurrence-free survival compared with placebo after the resection of primary gastrointestinal stromal tumour.
Funding: US National Institutes of Health and Novartis Pharmaceuticals.
Figures





Comment in
-
Adjuvant imatinib in GIST: a self-fulfilling prophecy, or more?Lancet. 2009 Mar 28;373(9669):1058-60. doi: 10.1016/S0140-6736(09)60562-6. Epub 2009 Mar 18. Lancet. 2009. PMID: 19303138 No abstract available.
References
-
- Nilsson B, Bumming P, Meis-Kindblom JM, et al. Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era--a population-based study in western Sweden. Cancer. 2005;103(4):821–829. - PubMed
-
- Tryggvason G, Gislason HG, Magnusson MK, Jonasson JG. Gastrointestinal stromal tumors in Iceland, 1990–2003: The Icelandic GIST study, a population-based incidence and pathologic risk stratification study. Int J Cancer. 2005 - PubMed
-
- Hirota S, Isozaki K, Moriyama Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279(5350):577–580. - PubMed
-
- Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21(23):4342–4349. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

