The effect of pharmaceutical patent term length on research and development and drug expenditures in Canada
- PMID: 19305720
- PMCID: PMC2585454
The effect of pharmaceutical patent term length on research and development and drug expenditures in Canada
Abstract
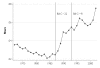
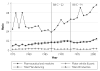
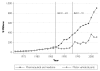
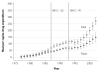
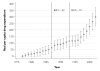
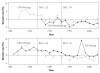
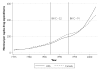
While pharmaceutical patent terms have increased in Canada, increases in patented drug spending have been mitigated by price controls and retrenchment of public prescription drug subsidy programs. We estimate the net effects of these offsetting policies on domestic pharmaceutical R&D expenditures and also provide an upper-bound estimate on the effects of these policies on Canadian pharmaceutical spending over the period 1988-2002. We estimate that R&D spending increased by $4.4 billion (1997 dollars). Drug spending increased by $3.9 billion at most and, quite likely, by much less. Cutbacks to public drug subsidies and the introduction of price controls likely mitigated drug spending growth. In cost-benefit terms, we suspect that the patent extension policies have been beneficial to Canada.
Même si la durée des brevets a augmenté au Canada, les augmentations dans les dépenses en médicaments brevetés ont été atténuées par les contrôles des prix et l’annulation de programmes de subventions pour médicaments sur ordonnance. Nous estimons les effets nets de ces politiques compensatoires sur les dépenses en R&D dans le domaine pharmaceutique à l’échelle nationale et fournissons également une estimation de la limite supérieure des effets de ces politiques sur les dépenses en médicaments de 1988 à 2002. Nous estimons que les dépenses en R&D ont augmenté de 4,4 G$ (dollars de 1997). Les dépenses en médicaments ont augmenté de 3,9 G$ au plus et, fort probablement, de beaucoup moins. Les réductions des subventions publiques aux programmes d’assurance-médicaments et, possiblement, l’introduction de contrôles des prix ont probablement réfréné les augmentations des dépenses en médicaments. Sur le plan des coûts-avantages, nous avons l’impression que les politiques sur la prolongation des brevets ont été bénéfiques pour le Canada.
Figures
References
-
- Anderson M. Mimeo. Queen’s University, Kingston, ON, Canada: 1997. Evergreening in the Canadian Pharmaceutical Industry.
-
- Anis A.H., Wen Q. Price Regulation of Pharmaceuticals in Canada. Journal of Health Economics. 1998;17:21–38. - PubMed
-
- Berndt E.R. Pharmaceuticals in US Health Care: Determinants of Quantity and Price. Journal of Economic Perspectives. 2002;16:45–66. - PubMed
LinkOut - more resources
Full Text Sources
