Use of the pharmacological inhibitor BX795 to study the regulation and physiological roles of TBK1 and IkappaB kinase epsilon: a distinct upstream kinase mediates Ser-172 phosphorylation and activation
- PMID: 19307177
- PMCID: PMC2682862
- DOI: 10.1074/jbc.M109.000414
Use of the pharmacological inhibitor BX795 to study the regulation and physiological roles of TBK1 and IkappaB kinase epsilon: a distinct upstream kinase mediates Ser-172 phosphorylation and activation
Abstract
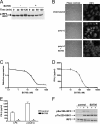
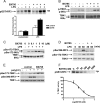
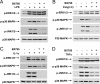
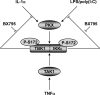
TANK-binding kinase 1 (TBK1) and IkappaB kinase epsilon (IKKepsilon) regulate the production of Type 1 interferons during bacterial and viral infection, but the lack of useful pharmacological inhibitors has hampered progress in identifying additional physiological roles of these protein kinases and how they are regulated. Here we demonstrate that BX795, a potent and relatively specific inhibitor of TBK1 and IKKepsilon, blocked the phosphorylation, nuclear translocation, and transcriptional activity of interferon regulatory factor 3 and, hence, the production of interferon-beta in macrophages stimulated with poly(I:C) or lipopolysaccharide (LPS). In contrast, BX795 had no effect on the canonical NFkappaB signaling pathway. Although BX795 blocked the autophosphorylation of overexpressed TBK1 and IKKepsilon at Ser-172 and, hence, the autoactivation of these protein kinases, it did not inhibit the phosphorylation of endogenous TBK1 and IKKepsilon at Ser-172 in response to LPS, poly(I:C), interleukin-1alpha (IL-1alpha), or tumor necrosis factor alpha and actually enhanced the LPS, poly(I:C), and IL-1alpha-stimulated phosphorylation of this residue. These results demonstrate that the phosphorylation of Ser-172 and the activation of TBK1 and IKKepsilon are catalyzed by a distinct protein kinase(s) in vivo and that TBK1 and IKKepsilon control a feedback loop that limits their activation by LPS, poly(I:C) and IL-1alpha (but not tumor necrosis factor alpha) to prevent the hyperactivation of these enzymes.
Figures






References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

