Indoleamine 2,3-dioxygenase overexpression causes kynurenine-modification of proteins, fiber cell apoptosis and cataract formation in the mouse lens
- PMID: 19308046
- PMCID: PMC2722445
- DOI: 10.1038/labinvest.2009.22
Indoleamine 2,3-dioxygenase overexpression causes kynurenine-modification of proteins, fiber cell apoptosis and cataract formation in the mouse lens
Abstract
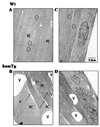
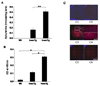
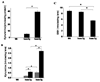
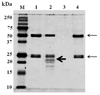
Indoleamine 2,3-dioxygenase (IDO) is the first enzyme in the kynurenine pathway. The kynurenines formed in this pathway chemically modify proteins and cause apoptosis in cells. Evidence suggests that kynurenines and their protein modifications are involved in cataract formation, but this has yet to be directly demonstrated. We generated transgenic (Tg) mouse lines that overexpress human IDO in the lens. Homozygous Tg (homTg) lenses had higher IDO immunoreactivity, approximately 4.5 times greater IDO mRNA, and approximately 8 times higher IDO activity compared to lenses from hemizygous Tg (hemTg) animals. The kynurenine content was threefold higher in homTg than in hemTg but was not detected in wild-type (Wt) lenses. Kynurenine modifications were approximately 2.6 times greater in homTg than in hemTg or Wt. HomTg lenses had vacuoles in the epithelium and cortical fiber cells. Kynurenine modifications coincided with apoptosis in the secondary fiber cells of homTg lenses. Caspase-3 and caspase-9 activities were markedly higher in homTg than in hemTg and Wt. The glutathione content was approximately 36% lower in homTg compared to hemTg and Wt lenses. HomTg animals also developed bilateral cataracts within 3 months of birth. Together these data demonstrate that IDO-mediated production of kynurenines results in defects in fiber cell differentiation and their apoptosis and suggest that IDO activity is kept low in the lens to prevent deleterious effects by kynurenines.
Figures







References
-
- King NJ, Thomas SR. Molecules in focus: indoleamine 2,3-dioxygenase. Int J Biochem Cell Biol. 2007;39:2167–2172. - PubMed
-
- Takikawa O. Biochemical and medical aspects of the indoleamine 2,3-dioxygenase-initiated L-tryptophan metabolism. Biochem Biophys Res Commun. 2005;338:12–19. - PubMed
-
- MacKenzie CR, Heseler K, Muller A, et al. Role of indoleamine 2,3-dioxygenase in antimicrobial defence and immuno-regulation: tryptophan depletion versus production of toxic kynurenines. Curr Drug Metab. 2007;8:237–244. - PubMed
-
- Fallarino F, Gizzi S, Mosci P, et al. Tryptophan catabolism in IDO+ plasmacytoid dendritic cells. Curr Drug Metab. 2007;8:209–216. - PubMed
-
- Mellor AL, Munn D, Chandler P, et al. Tryptophan catabolism and T cell responses. Adv Exp Med Biol. 2003;527:27–35. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

