Caveolin-1 regulates the antagonistic pleiotropic properties of cellular senescence through a novel Mdm2/p53-mediated pathway
- PMID: 19318577
- PMCID: PMC2692066
- DOI: 10.1158/0008-5472.CAN-08-2857
Caveolin-1 regulates the antagonistic pleiotropic properties of cellular senescence through a novel Mdm2/p53-mediated pathway
Abstract
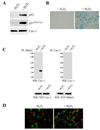
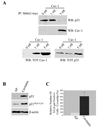
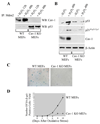
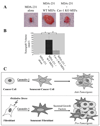
We show that caveolin-1 is a novel binding protein for Mdm2. After oxidative stress, caveolin-1 sequesters Mdm2 away from p53, leading to stabilization of p53 and up-regulation of p21(Waf1/Cip1) in human fibroblasts. Expression of a peptide corresponding to the Mdm2 binding domain of caveolin-1 is sufficient to up-regulate p53 and p21(Waf1/Cip1) protein expression and induce premature senescence. Oxidative stress-induced activation of the p53/p21(Waf1/Cip1) pathway and induction of premature senescence are compromised in caveolin-1 null mouse embryonic fibroblasts (MEF). We also show that reintroduction of caveolin-1 in oncogenic Ras (Ras(G12V))-transformed fibroblasts, which express residual levels of caveolin-1, is sufficient to promote cellular senescence. Moreover, caveolin-1 expression in MEFs is required for senescent fibroblast-induced stimulation of cell growth and tumorigenesis of both Ras(G12V)-transformed fibroblasts and MDA-MB-231 breast cancer epithelial cells both in vitro and in vivo. Thus, our results propose caveolin-1 as a key mediator of the antagonistic pleiotropic properties of cellular senescence.
Figures


References
-
- Williams GC. Pleiotropy, natural selection, and the evolution of senescence. Evolution. 1957;11:398–411.
-
- Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005;120:513–522. - PubMed
-
- Campisi J. Cellular senescence as a tumor-suppressor mechanism. Trends Cell Biol. 2001;11:S27–S31. - PubMed
-
- Millis AJ, Hoyle M, McCue HM, Martini H. Differential expression of metalloproteinase and tissue inhibitor of metalloproteinase genes in aged human fibroblasts. Exp Cell Res. 1992;201:373–379. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

