Improved survival in rhesus macaques immunized with modified vaccinia virus Ankara recombinants expressing simian immunodeficiency virus envelope correlates with reduction in memory CD4+ T-cell loss and higher titers of neutralizing antibody
- PMID: 19321617
- PMCID: PMC2681965
- DOI: 10.1128/JVI.02598-08
Improved survival in rhesus macaques immunized with modified vaccinia virus Ankara recombinants expressing simian immunodeficiency virus envelope correlates with reduction in memory CD4+ T-cell loss and higher titers of neutralizing antibody
Abstract
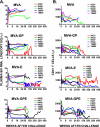
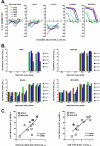
Previous studies demonstrated that immunization of macaques with simian immunodeficiency virus (SIV) Gag-Pol and Env recombinants of the attenuated poxvirus modified vaccinia virus Ankara (MVA) provided protection from high viremia and AIDS following challenge with a pathogenic strain of SIV. Although all animals became infected, plasma viremia was significantly reduced in animals that received the MVA-SIV recombinant vaccines compared with animals that received nonrecombinant MVA. Most importantly, the reduction in viremia resulted in a significant increase in median and cumulative survival. Continued analysis of these animals over the subsequent 9 years has shown that they maintain a survival advantage, although all but two of the macaques have progressed to AIDS. Importantly, improved survival correlated with preservation of memory CD4(+) T cells in the peripheral blood. The greatest survival advantage was observed in macaques immunized with regimens containing SIV Env, and the titer of neutralizing antibodies to the challenge virus prior to or shortly following challenge correlated with preservation of CD4(+) T cells. These data are consistent with a role for neutralizing antibodies in nonsterilizing protection from high viremia and associated memory CD4(+) T-cell loss.
Figures



Similar articles
-
Comparative efficacy of recombinant modified vaccinia virus Ankara expressing simian immunodeficiency virus (SIV) Gag-Pol and/or Env in macaques challenged with pathogenic SIV.J Virol. 2000 Mar;74(6):2740-51. doi: 10.1128/jvi.74.6.2740-2751.2000. J Virol. 2000. PMID: 10684290 Free PMC article.
-
Recombinant modified vaccinia virus ankara expressing the surface gp120 of simian immunodeficiency virus (SIV) primes for a rapid neutralizing antibody response to SIV infection in macaques.J Virol. 2000 Mar;74(6):2960-5. doi: 10.1128/jvi.74.6.2960-2965.2000. J Virol. 2000. PMID: 10684319 Free PMC article.
-
Virus-Like Particles Displaying Trimeric Simian Immunodeficiency Virus (SIV) Envelope gp160 Enhance the Breadth of DNA/Modified Vaccinia Virus Ankara SIV Vaccine-Induced Antibody Responses in Rhesus Macaques.J Virol. 2016 Sep 12;90(19):8842-54. doi: 10.1128/JVI.01163-16. Print 2016 Oct 1. J Virol. 2016. PMID: 27466414 Free PMC article.
-
Host range restricted, non-replicating vaccinia virus vectors as vaccine candidates.Adv Exp Med Biol. 1996;397:7-13. doi: 10.1007/978-1-4899-1382-1_2. Adv Exp Med Biol. 1996. PMID: 8718576 Free PMC article. Review.
-
Memory T cells in Rhesus macaques.Adv Exp Med Biol. 2010;684:126-44. doi: 10.1007/978-1-4419-6451-9_10. Adv Exp Med Biol. 2010. PMID: 20795545 Free PMC article. Review.
Cited by
-
Recombinant rubella vectors elicit SIV Gag-specific T cell responses with cytotoxic potential in rhesus macaques.Vaccine. 2015 Apr 27;33(18):2167-74. doi: 10.1016/j.vaccine.2015.02.067. Epub 2015 Mar 21. Vaccine. 2015. PMID: 25802183 Free PMC article.
-
Significant protection against high-dose simian immunodeficiency virus challenge conferred by a new prime-boost vaccine regimen.J Virol. 2011 Jun;85(12):5764-72. doi: 10.1128/JVI.00342-11. Epub 2011 Apr 13. J Virol. 2011. PMID: 21490100 Free PMC article.
-
A multivalent and cross-protective vaccine strategy against arenaviruses associated with human disease.PLoS Pathog. 2009 Dec;5(12):e1000695. doi: 10.1371/journal.ppat.1000695. Epub 2009 Dec 18. PLoS Pathog. 2009. PMID: 20019801 Free PMC article.
-
Heterogeneity in neutralization sensitivities of viruses comprising the simian immunodeficiency virus SIVsmE660 isolate and vaccine challenge stock.J Virol. 2013 May;87(10):5477-92. doi: 10.1128/JVI.03419-12. Epub 2013 Mar 6. J Virol. 2013. PMID: 23468494 Free PMC article.
-
Early short-term antiretroviral therapy is associated with a reduced prevalence of CD8(+)FoxP3(+) T cells in simian immunodeficiency virus-infected controller rhesus macaques.AIDS Res Hum Retroviruses. 2011 Jul;27(7):763-75. doi: 10.1089/AID.2010.0251. Epub 2011 Jan 17. AIDS Res Hum Retroviruses. 2011. PMID: 21142402 Free PMC article.
References
-
- Amara, R. R., C. Ibegbu, F. Villinger, D. C. Montefiori, S. Sharma, P. Nigam, Y. Xu, H. M. McClure, and H. L. Robinson. 2005. Studies using a viral challenge and CD8 T cell depletions on the roles of cellular and humoral immunity in the control of an SHIV-89.6P challenge in DNA/MVA-vaccinated macaques. Virology 343246-255. - PubMed
-
- Amara, R. R., J. M. Smith, S. I. Staprans, D. C. Montefiori, F. Villinger, J. D. Altman, S. P. O'Neil, N. L. Kozyr, Y. Xu, L. S. Wyatt, P. L. Earl, J. G. Herndon, J. M. McNicholl, H. M. McClure, B. Moss, and H. L. Robinson. 2002. Critical role for Env as well as Gag-Pol in control of a simian-human immunodeficiency virus 89.6P challenge by a DNA prime/recombinant modified vaccinia virus Ankara vaccine. J. Virol. 766138-6146. - PMC - PubMed
-
- Amara, R. R., F. Villinger, J. D. Altman, S. L. Lydy, S. P. O'Neil, S. I. Staprans, D. C. Montefiori, Y. Xu, J. G. Herndon, L. S. Wyatt, M. A. Candido, N. L. Kozyr, P. L. Earl, J. M. Smith, H. L. Ma, B. D. Grimm, M. L. Hulsey, J. Miller, H. M. McClure, J. M. McNicholl, B. Moss, and H. L. Robinson. 2001. Control of a mucosal challenge and prevention of AIDS by a multiprotein DNA/MVA vaccine. Science 29269-74. - PubMed
-
- Baba, T. W., V. Liska, R. Hofmann-Lehmann, J. Vlasak, W. Xu, S. Ayehunie, L. A. Cavacini, M. R. Posner, H. Katinger, G. Stiegler, B. J. Bernacky, T. A. Rizvi, R. Schmidt, L. R. Hill, M. E. Keeling, Y. Lu, J. E. Wright, T. C. Chou, and R. M. Ruprecht. 2000. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian-human immunodeficiency virus infection. Nat. Med. 6200-206. - PubMed
-
- Barnett, S. W., I. K. Srivastava, E. Kan, F. Zhou, A. Goodsell, A. D. Cristillo, M. G. Ferrai, D. E. Weiss, N. L. Letvin, D. Montefiori, R. Pal, and M. Vajdy. 2008. Protection of macaques against vaginal SHIV challenge by systemic or mucosal and systemic vaccinations with HIV-envelope. AIDS 22339-348. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

