Raphé neurons stimulate respiratory circuit activity by multiple mechanisms via endogenously released serotonin and substance P
- PMID: 19321769
- PMCID: PMC2940110
- DOI: 10.1523/JNEUROSCI.5271-08.2009
Raphé neurons stimulate respiratory circuit activity by multiple mechanisms via endogenously released serotonin and substance P
Abstract
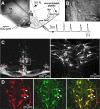
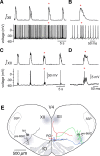
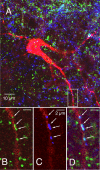
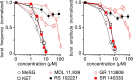
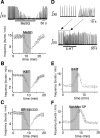
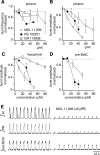
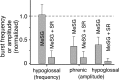
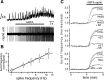
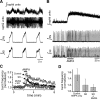
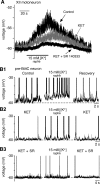
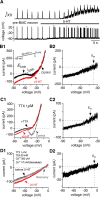
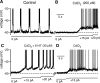
Brainstem serotonin (5-HT) neurons modulate activity of many neural circuits in the mammalian brain, but in many cases endogenous mechanisms have not been resolved. Here, we analyzed actions of raphé 5-HT neurons on respiratory network activity including at the level of the pre-Bötzinger complex (pre-BötC) in neonatal rat medullary slices in vitro, and in the more intact nervous system of juvenile rats in arterially perfused brainstem-spinal cord preparations in situ. At basal levels of activity, excitation of the respiratory network via simultaneous release of 5-HT and substance P (SP), acting at 5-HT(2A/2C), 5-HT(4), and/or neurokinin-1 receptors, was required to maintain inspiratory motor output in both the neonatal and juvenile systems. The midline raphé obscurus contained spontaneously active 5-HT neurons, some of which projected to the pre-BötC and hypoglossal motoneurons, colocalized 5-HT and SP, and received reciprocal excitatory connections from the pre-BötC. Experimentally augmenting raphé obscurus activity increased motor output by simultaneously exciting pre-BötC and motor neurons. Biophysical analyses in vitro demonstrated that 5-HT and SP modulated background cation conductances in pre-BötC and motor neurons, including a nonselective cation leak current that contributed to the resting potential, which explains the neuronal depolarization that augmented motor output. Furthermore, we found that 5-HT, but not SP, can transform the electrophysiological phenotype of some pre-BötC neurons to intrinsic bursters, providing 5-HT with an additional role in promoting rhythm generation. We conclude that raphé 5-HT neurons excite key circuit components required for generation of respiratory motor output.
Figures


References
-
- Al-Zubaidy ZA, Erickson RL, Greer JJ. Serotonergic and noradrenergic effects on respiratory neural discharge in the medullary slice preparation of neonatal rats. Pflugers Arch. 1996;431:942–949. - PubMed
-
- Bayliss DA, Li YW, Talley EM. Effects of serotonin on caudal raphé neurons: inhibition of N- and P/Q-type calcium channels and the afterhyperpolarization. J Neurophysiol. 1997;77:1362–1374. - PubMed
-
- Bobker DH, Williams JT. Serotonin augments the cation current Ih in central neurons. Neuron. 1989;2:1535–1540. - PubMed
-
- Butera RJ, Jr, Rinzel J, Smith JC. Models of respiratory rhythm generation in the pre-Bötzinger complex. I. Bursting pacemaker neurons. J Neurophysiol. 1999a;82:382–397. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
