Floral and insect-induced volatile formation in Arabidopsis lyrata ssp. petraea, a perennial, outcrossing relative of A. thaliana
- PMID: 19322583
- PMCID: PMC2687518
- DOI: 10.1007/s00425-009-0921-7
Floral and insect-induced volatile formation in Arabidopsis lyrata ssp. petraea, a perennial, outcrossing relative of A. thaliana
Abstract
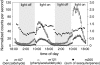
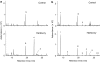
Volatile organic compounds have been reported to serve some important roles in plant communication with other organisms, but little is known about the biological functions of most of these substances. To gain insight into this problem, we have compared differences in floral and vegetative volatiles between two closely related plant species with different life histories. The self-pollinating annual, Arabidopsis thaliana, and its relative, the outcrossing perennial, Arabidopsis lyrata, have markedly divergent life cycles and breeding systems. We show that these differences are in part reflected in the formation of distinct volatile mixtures in flowers and foliage. Volatiles emitted from flowers of a German A. lyrata ssp. petraea population are dominated by benzenoid compounds in contrast to the previously described sesquiterpene-dominated emissions of A. thaliana flowers. Flowers of A. lyrata ssp. petraea release benzenoid volatiles in a diurnal rhythm with highest emission rates at midday coinciding with observed visitations of pollinating insects. Insect feeding on leaves of A. lyrata ssp. petraea causes a variable release of the volatiles methyl salicylate, C11- and C16-homoterpenes, nerolidol, plus the sesquiterpene (E)-beta-caryophyllene, which in A. thaliana is emitted exclusively from flowers. An insect-induced gene (AlCarS) with high sequence similarity to the florally expressed (E)-beta-caryophyllene synthase (AtTPS21) from A. thaliana was identified from individuals of a German A. lyrata ssp. petraea population. Recombinant AlCarS converts the sesquiterpene precursor, farnesyl diphosphate, into (E)-beta-caryophyllene with alpha-humulene and alpha-copaene as minor products indicating its close functional relationship to the A. thaliana AtTPS21. Differential regulation of these genes in flowers and foliage is consistent with the different functions of volatiles in the two Arabidopsis species.
Figures
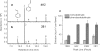




References
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1038/hdy.1989.56', 'is_inner': False, 'url': 'https://doi.org/10.1038/hdy.1989.56'}]}
- Abbott RJ, Gomes MF (1989) Population genetic structure and outcrossing rate of Arabidopsis thaliana (L) Heynh. Heredity 62:411–418
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1007/s00438-002-0709-y', 'is_inner': False, 'url': 'https://doi.org/10.1007/s00438-002-0709-y'}, {'type': 'PubMed', 'value': '12207221', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/12207221/'}]}
- Aubourg S, Lecharny A, Bohlmann J (2002) Genomic analysis of the terpenoid synthase (AtTPS) gene family of Arabidopsis thaliana. Mol Genet Genomics 267:730–745 - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1105/tpc.12.7.1093', 'is_inner': False, 'url': 'https://doi.org/10.1105/tpc.12.7.1093'}, {'type': 'PMC', 'value': 'PMC149051', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC149051/'}, {'type': 'PubMed', 'value': '10899976', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/10899976/'}]}
- Blanc G, Barakat A, Guyot R, Cooke R, Delseny I (2000) Extensive duplication and reshuffling in the Arabidopsis genome. Plant Cell 12:1093–1101 - PMC - PubMed
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1023/B:JOEC.0000006446.21160.c1', 'is_inner': False, 'url': 'https://doi.org/10.1023/b:joec.0000006446.21160.c1'}]}
- Blight MM, LeMetayer M, Delegue MHP, Pickett JA, Marion-Poll F, Wadhams LJ (1997) Identification of floral volatiles involved in recognition of oilseed rape flowers, Brassica napus by honeybees, Apis mellifera. J Chem Ecol 23:1715–1727
-
- {'text': '', 'ref_index': 1, 'ids': [{'type': 'DOI', 'value': '10.1105/tpc.007989', 'is_inner': False, 'url': 'https://doi.org/10.1105/tpc.007989'}, {'type': 'PMC', 'value': 'PMC141215', 'is_inner': False, 'url': 'https://pmc.ncbi.nlm.nih.gov/articles/PMC141215/'}, {'type': 'PubMed', 'value': '12566586', 'is_inner': True, 'url': 'https://pubmed.ncbi.nlm.nih.gov/12566586/'}]}
- Chen F, Tholl D, D’Auria JC, Farooq A, Pichersky E, Gershenzon J (2003) Biosynthesis and emission of terpenoid volatiles from Arabidopsis flowers. Plant Cell 15:481–494 - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources

