Transforming growth factor-beta 1 induces intestinal myofibroblast differentiation and modulates their migration
- PMID: 19322915
- PMCID: PMC2665137
- DOI: 10.3748/wjg.15.1431
Transforming growth factor-beta 1 induces intestinal myofibroblast differentiation and modulates their migration
Abstract
Aim: To investigate the effects of transforming growth factor beta 1 (TGF-beta 1) on the differentiation of colonic lamina propria fibroblasts (CLPF) into myofibroblasts in vitro.
Methods: Primary CLPF cultures were incubated with TGF-beta 1 and analyzed for production of alpha-smooth muscle actin (alpha-SMA), fibronectin (FN) and FN isoforms. Migration assays were performed in a modified 48-well Boyden chamber. Levels of total and phosphorylated focal adhesion kinase (FAK) in CLPF were analyzed after induction of migration.
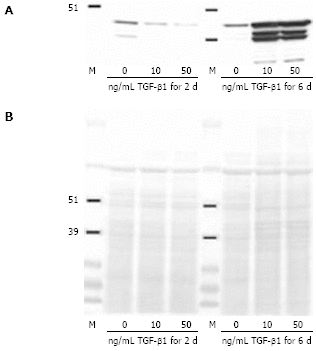
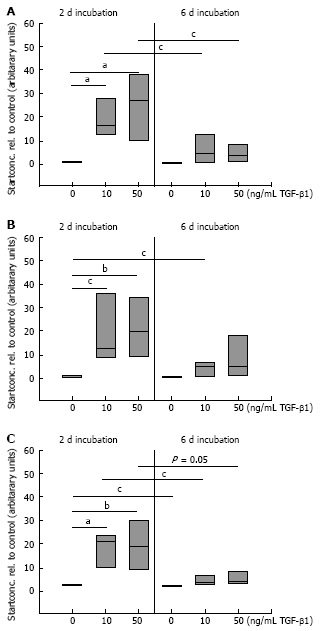
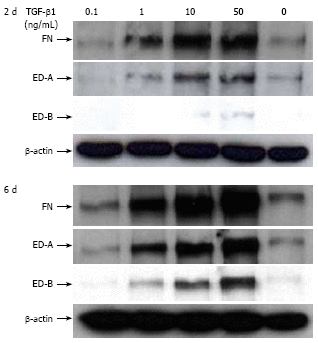
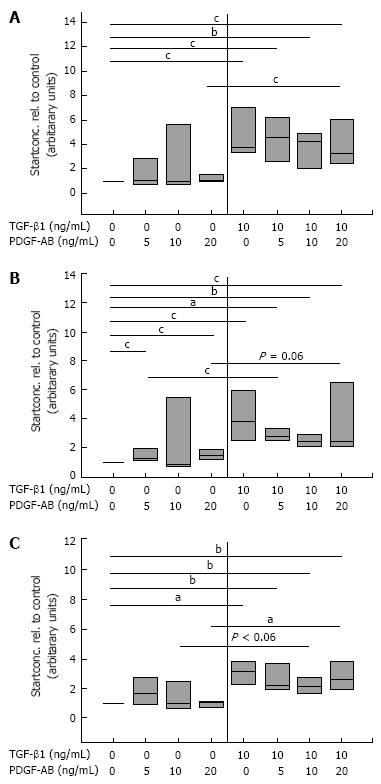
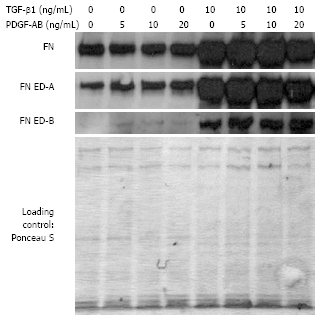
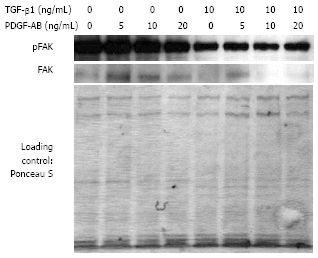
Results: Incubation of CLPF with TGF-beta 1 for 2 d did not change alpha-SMA levels, while TGF-beta 1 treatment for 6 d significantly increased alpha-SMA production. Short term incubation (6 h) with TGF-beta 1 enhanced CLPF migration, while long term treatment (6 d) of CLPF with TGF-beta 1 reduced migration to 15%-37% compared to untreated cells. FN and FN isoform mRNA expression were increased after short term incubation with TGF-beta 1 (2 d) in contrast to long term incubation with TGF-beta 1 for 6 d. After induction of migration, TGF-beta 1-preincubated CLPF showed higher amounts of FN and its isoforms and lower levels of total and phosphorylated FAK than untreated cells.
Conclusion: Long term incubation of CLPF with TGF-beta 1 induced differentiation into myofibroblasts with enhanced alpha-SMA, reduced migratory potential and FAK phosphorylation, and increased FN production. In contrast, short term contact (6 h) of fibroblasts with TGF-beta 1 induced a dose-dependent increase of cell migration and FAK phosphorylation without induction of alpha-SMA production.
Figures


Similar articles
-
Inflammation modulates fibronectin isoform expression in colonic lamina propria fibroblasts (CLPF).Int J Colorectal Dis. 2008 Oct;23(10):947-55. doi: 10.1007/s00384-008-0523-z. Epub 2008 Jul 17. Int J Colorectal Dis. 2008. PMID: 18633626
-
Autocrine fibronectin-induced migration of human colonic fibroblasts.Am J Gastroenterol. 2004 Feb;99(2):335-40. doi: 10.1111/j.1572-0241.2004.04044.x. Am J Gastroenterol. 2004. PMID: 15046226
-
Reduced migration of fibroblasts in inflammatory bowel disease: role of inflammatory mediators and focal adhesion kinase.Gastroenterology. 2003 Nov;125(5):1341-54. doi: 10.1016/j.gastro.2003.07.004. Gastroenterology. 2003. PMID: 14598250
-
Evidence for a differential expression of fibronectin splice forms ED-A and ED-B in Crohn's disease (CD) mucosa.Int J Colorectal Dis. 2007 Jun;22(6):611-23. doi: 10.1007/s00384-006-0188-4. Epub 2006 Nov 30. Int J Colorectal Dis. 2007. PMID: 17136547
-
Prostaglandin E2 inhibits migration of colonic lamina propria fibroblasts.Inflamm Bowel Dis. 2010 Sep;16(9):1505-13. doi: 10.1002/ibd.21255. Inflamm Bowel Dis. 2010. PMID: 20803697
Cited by
-
Asthmatic Eosinophils Alter the Gene Expression of Extracellular Matrix Proteins in Airway Smooth Muscle Cells and Pulmonary Fibroblasts.Int J Mol Sci. 2022 Apr 7;23(8):4086. doi: 10.3390/ijms23084086. Int J Mol Sci. 2022. PMID: 35456903 Free PMC article.
-
Vascular endothelial growth factor-D mediates fibrogenic response in myofibroblasts.Mol Cell Biochem. 2016 Feb;413(1-2):127-35. doi: 10.1007/s11010-015-2646-1. Epub 2016 Jan 2. Mol Cell Biochem. 2016. PMID: 26724950
-
CLIC4 regulates TGF-β-dependent myofibroblast differentiation to produce a cancer stroma.Oncogene. 2014 Feb 13;33(7):842-50. doi: 10.1038/onc.2013.18. Epub 2013 Feb 18. Oncogene. 2014. PMID: 23416981 Free PMC article.
-
Physiological and pathological role of local and immigrating colonic stem cells.World J Gastroenterol. 2012 Jan 28;18(4):295-301. doi: 10.3748/wjg.v18.i4.295. World J Gastroenterol. 2012. PMID: 22294835 Free PMC article. Review.
-
MiR-155 modulates the inflammatory phenotype of intestinal myofibroblasts by targeting SOCS1 in ulcerative colitis.Exp Mol Med. 2015 May 22;47(5):e164. doi: 10.1038/emm.2015.21. Exp Mol Med. 2015. PMID: 25998827 Free PMC article.
References
-
- Kay EP, Lee MS, Seong GJ, Lee YG. TGF-beta s stimulate cell proliferation via an autocrine production of FGF-2 in corneal stromal fibroblasts. Curr Eye Res. 1998;17:286–293. - PubMed
-
- Strutz F, Zeisberg M, Renziehausen A, Raschke B, Becker V, van Kooten C, Müller G. TGF-beta 1 induces proliferation in human renal fibroblasts via induction of basic fibroblast growth factor (FGF-2) Kidney Int. 2001;59:579–592. - PubMed
-
- Thannickal VJ, Aldweib KD, Rajan T, Fanburg BL. Upregulated expression of fibroblast growth factor (FGF) receptors by transforming growth factor-beta1 (TGF-beta1) mediates enhanced mitogenic responses to FGFs in cultured human lung fibroblasts. Biochem Biophys Res Commun. 1998;251:437–441. - PubMed
-
- Adelmann-Grill BC, Wach F, Cully Z, Hein R, Krieg T. Chemotactic migration of normal dermal fibroblasts towards epidermal growth factor and its modulation by platelet-derived growth factor and transforming growth factor-beta. Eur J Cell Biol. 1990;51:322–326. - PubMed
-
- Andresen JL, Ehlers N. Chemotaxis of human keratocytes is increased by platelet-derived growth factor-BB, epidermal growth factor, transforming growth factor-alpha, acidic fibroblast growth factor, insulin-like growth factor-I, and transforming growth factor-beta. Curr Eye Res. 1998;17:79–87. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

