Patient-assessed versus physician-assessed disease severity and outcome in patients with nonspecific pain associated with major depressive disorder
- PMID: 19333404
- PMCID: PMC2660158
- DOI: 10.4088/pcc.08m00670
Patient-assessed versus physician-assessed disease severity and outcome in patients with nonspecific pain associated with major depressive disorder
Abstract
Objectives: This post hoc analysis compared how patients and physicians estimate disease severity and global improvement during 8 weeks of treatment for major depressive disorder (MDD) with associated nonspecific pain. In addition, predictors of pain and depression were identified.
Method: Data were derived from a double-blind, placebo-controlled, multicenter, European study (conducted from May 2005 to May 2006) in adult outpatients with MDD (DSM-IV criteria) and moderate pain not attributable to a diagnosed organic pain syndrome (Brief Pain Inventory-Short Form [BPI-SF] average pain score ≥ 3). Patients were randomly assigned to duloxetine 60 mg/day or placebo and treated for 8 weeks. Physicians were asked to rate severity of depression by using the Montgomery-Asberg Depression Rating Scale (MADRS) and the Clinical Global Impressions-Severity of Illness (CGI-S) and CGI-Improvement (CGI-I) scales. Patients were asked to assess pain using the BPI-SF, psychological symptomatology (9 domains including depression) with the Symptom Checklist-90-Revised (SCL-90-R), and overall improvement with the Patient Global Impression of Improvement (PGI-I). Multivariate linear regressions were performed as post hoc analyses to identify predictors of disease assessment at baseline and at the end of the study using a last-observation-carried-forward approach.
Results: All SCL-90-R domains improved during the 8 weeks of treatment. At baseline, the MADRS was associated only with the SCL-90-R obsessive-compulsive score, while the SCL-90-R depression score was associated with the BPI-SF average pain score and with many SCL-90-R subscores. The global impression of improvement was rated higher by the physicians than by the patients. At the end of the study, CGI-I was significantly associated with a decrease in depression severity (MADRS; p < .0001), younger age (p = .0005), and a decrease of the SCL-90-R interpersonal sensitivity score (p = .0359), but not with BPI-SF average pain. In contrast, patient-rated PGI-I was significantly associated with the SCL-90-R depressive domain (p < .0001), BPI-SF average pain (p = .0003), and the SCL-90-R anxiety domain (p = .0041) scores.
Conclusion: In patients with MDD associated with at least moderate nonspecific pain, physicians consider mainly the change in depressive symptoms as measured by MADRS in their CGI-I ratings, while patients also consider pain, depression, and anxiety in their PGI-I ratings. When treating depression and assessing treatment outcome, a broad spectrum of symptoms needs to be monitored.
Trial registration: clinicaltrials.gov Identifier: NCT00191919.
Figures
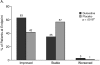
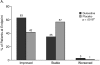
Similar articles
-
Efficacy and safety of duloxetine 60 mg once daily in the treatment of pain in patients with major depressive disorder and at least moderate pain of unknown etiology: a randomized controlled trial.J Clin Psychiatry. 2007 Nov;68(11):1707-16. doi: 10.4088/jcp.v68n1110. J Clin Psychiatry. 2007. PMID: 18052564 Clinical Trial.
-
Time course of improvement of different symptom clusters in patients with major depression and pain treated with duloxetine or placebo.Curr Med Res Opin. 2012 Jan;28(1):41-8. doi: 10.1185/03007995.2011.645561. Epub 2011 Dec 22. Curr Med Res Opin. 2012. PMID: 22126422
-
Duloxetine versus placebo in the treatment of major depressive disorder and associated painful physical symptoms: a replication study.Curr Med Res Opin. 2011 Oct;27(10):1859-67. doi: 10.1185/03007995.2011.609540. Epub 2011 Aug 12. Curr Med Res Opin. 2011. PMID: 21838410 Clinical Trial.
-
Novel Augmentation Strategies in Major Depression.Dan Med J. 2017 Apr;64(4):B5338. Dan Med J. 2017. PMID: 28385173 Review.
-
Patterns of depressive symptom response in duloxetine-treated outpatients with mild, moderate or more severe depression.Int J Clin Pract. 2007 Aug;61(8):1337-48. doi: 10.1111/j.1742-1241.2007.01444.x. Int J Clin Pract. 2007. PMID: 17627710 Review.
Cited by
-
Self-reported indications for antidepressant use in a population-based cohort of middle-aged and elderly.Int J Clin Pharm. 2016 Oct;38(5):1311-7. doi: 10.1007/s11096-016-0371-9. Epub 2016 Sep 1. Int J Clin Pharm. 2016. PMID: 27586370 Free PMC article.
-
A double-blind, randomized, placebo-controlled study assessing the efficacy and tolerability of desvenlafaxine 10 and 50 mg/day in adult outpatients with major depressive disorder.BMC Psychiatry. 2013 Mar 22;13:94. doi: 10.1186/1471-244X-13-94. BMC Psychiatry. 2013. PMID: 23517291 Free PMC article. Clinical Trial.
-
Do general practitioners and psychiatrists agree about defining cure from depression? The DEsCRIBE™ survey.BMC Psychiatry. 2011 Oct 14;11:169. doi: 10.1186/1471-244X-11-169. BMC Psychiatry. 2011. PMID: 21999407 Free PMC article.
-
Connectivity-guided intermittent theta burst versus repetitive transcranial magnetic stimulation for treatment-resistant depression: a randomized controlled trial.Nat Med. 2024 Feb;30(2):403-413. doi: 10.1038/s41591-023-02764-z. Epub 2024 Jan 16. Nat Med. 2024. PMID: 38228914 Free PMC article. Clinical Trial.
-
Relative effectiveness and adverse effects of cervical manipulation, mobilisation and the activator instrument in patients with sub-acute non-specific neck pain: results from a stopped randomised trial.Chiropr Osteopat. 2010 Jul 9;18:20. doi: 10.1186/1746-1340-18-20. Chiropr Osteopat. 2010. PMID: 20618936 Free PMC article.
References
-
- Guy W. Rockville, Md: National Institute of Mental Health; 1976. ECDEU Assessment Manual for Psychopharmacology. US Dept Health, Education, and Welfare publication (ADM) 76-338; pp. 218–222.
-
- Lehman AF, Babigian HM, Reed SK. The epidemiology of treatment for chronic and nonchronic mental disorders. J Nerv Ment Dis. 1984;172:658–666. - PubMed
-
- Beneke M, Rasmus W. “Clinical Global Impressions” (ECDEU): some critical comments. Pharmacopsychiatry. 1991;25(4):171–176. - PubMed
-
- Leon AC, Shear MK, Klerman GL, et al. A comparison of symptom determinants of patient and clinician global ratings in patients with panic disorder and depression. J Clin Psychopharmacol. 1993;13:327–331. - PubMed
-
- Zaider TI, Heimberg RG, Fresco DM, et al. Evaluation of the Clinical Global Impressions scale among individuals with social anxiety disorder. Psychol Med. 2003;33:611–622. - PubMed
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
