Convergent, not serial, striatal and pallidal circuits regulate opioid-induced food intake
- PMID: 19336249
- PMCID: PMC2699890
- DOI: 10.1016/j.neuroscience.2009.03.057
Convergent, not serial, striatal and pallidal circuits regulate opioid-induced food intake
Abstract
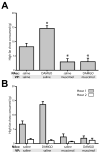
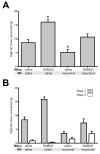
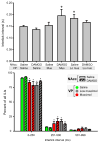
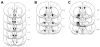
Mu opioid receptor (MOR) signaling in the nucleus accumbens (NAcc) elicits marked increases in the consumption of palatable tastants. However, the mechanism and circuitry underlying this effect are not fully understood. Multiple downstream target regions have been implicated in mediating this effect but the role of the ventral pallidum (VP), a primary target of NAcc efferents, has not been well defined. To probe the mechanisms underlying increased consumption, we identified behavioral changes in rats' licking patterns following NAcc MOR stimulation. Because the temporal structure of licking reflects the physiological substrates modulating consumption, these measures provide a useful tool in dissecting the cause of increased consumption following NAcc MOR stimulation. Next, we used a combination of pharmacological inactivation and lesions to define the role of the VP in hyperphagia following infusion of the MOR-specific agonist [D-Ala2, N-MePhe4, Gly-ol]-enkephalin (DAMGO) into the NAcc. In agreement with previous studies, results from lick microstructure analysis suggest that NAcc MOR stimulation augments intake through a palatability-driven mechanism. Our results also demonstrate an important role for the VP in normal feeding behavior: pharmacological inactivation of the VP suppresses baseline and NAcc DAMGO-induced consumption. However, this interaction does not occur through a serial circuit requiring direct projections from the NAcc to the VP. Rather, our results indicate that NAcc and VP circuits converge on a common downstream target that regulates food intake.
Figures





Similar articles
-
mu-Opioid receptor stimulation in the nucleus accumbens elevates fatty tastant intake by increasing palatability and suppressing satiety signals.Am J Physiol Regul Integr Comp Physiol. 2011 Jul;301(1):R244-54. doi: 10.1152/ajpregu.00406.2010. Epub 2011 May 4. Am J Physiol Regul Integr Comp Physiol. 2011. PMID: 21543633 Free PMC article.
-
Modulation of feeding and locomotion through mu and delta opioid receptor signaling in the nucleus accumbens.Neuropeptides. 2010 Jun;44(3):225-32. doi: 10.1016/j.npep.2009.12.002. Epub 2009 Dec 30. Neuropeptides. 2010. PMID: 20044138 Free PMC article.
-
Accumbal core: essential link in feed-forward spiraling striato-nigro-striatal in series connected loop.Neuroscience. 2013 Nov 12;252:60-7. doi: 10.1016/j.neuroscience.2013.07.066. Epub 2013 Aug 8. Neuroscience. 2013. PMID: 23933312
-
Nucleus accumbens opioids regulate flavor-based preferences in food consumption.Neuroscience. 2006 Nov 17;143(1):309-17. doi: 10.1016/j.neuroscience.2006.06.067. Neuroscience. 2006. PMID: 17049180
-
gamma-Aminobutyric acid and mu-opioid receptor localization and adaptation in the basal forebrain.Adv Exp Med Biol. 1991;295:101-17. doi: 10.1007/978-1-4757-0145-6_3. Adv Exp Med Biol. 1991. PMID: 1663697 Review.
Cited by
-
Altered hypothalamic response to food in smokers.Am J Clin Nutr. 2013 Jan;97(1):15-22. doi: 10.3945/ajcn.112.043307. Epub 2012 Dec 12. Am J Clin Nutr. 2013. PMID: 23235196 Free PMC article.
-
Mu opioid receptor antagonism in the nucleus accumbens shell blocks consumption of a preferred sucrose solution in an anticipatory contrast paradigm.Neuroscience. 2014 Mar 7;261:144-52. doi: 10.1016/j.neuroscience.2013.12.004. Epub 2013 Dec 14. Neuroscience. 2014. PMID: 24342569 Free PMC article.
-
Advances in the neurobiological bases for food 'liking' versus 'wanting'.Physiol Behav. 2014 Sep;136:22-30. doi: 10.1016/j.physbeh.2014.05.022. Epub 2014 May 27. Physiol Behav. 2014. PMID: 24874776 Free PMC article. Review.
-
The ventral pallidum: Subregion-specific functional anatomy and roles in motivated behaviors.Prog Neurobiol. 2015 Jul;130:29-70. doi: 10.1016/j.pneurobio.2015.03.005. Epub 2015 Apr 6. Prog Neurobiol. 2015. PMID: 25857550 Free PMC article. Review.
-
Long-term changes in reward-seeking following morphine withdrawal are associated with altered N-methyl-D-aspartate receptor 1 splice variants in the amygdala.Neuroscience. 2012 Oct 25;223:45-55. doi: 10.1016/j.neuroscience.2012.07.037. Epub 2012 Aug 2. Neuroscience. 2012. PMID: 22863572 Free PMC article.
References
-
- Adam TC, Epel ES. Stress, eating and the reward system. Physiol Behav. 2007;91:449–458. - PubMed
-
- Alheid GF. Extended amygdala and basal forebrain. Ann N Y Acad Sci. 2003;985:185–205. - PubMed
-
- Austin MC, Kalivas PW. Blockade of enkephalinergic and GABAergic mediated locomotion in the nucleus accumbens by muscimol in the ventral pallidum. Jpn J Pharmacol. 1989;50:487–490. - PubMed
-
- Baird JP, St John SJ, Nguyen EA. Temporal and qualitative dynamics of conditioned taste aversion processing: combined generalization testing and licking microstructure analysis. Behav Neurosci. 2005;119:983–1003. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

