Achieving lipid goals with rosuvastatin compared with simvastatin in high risk patients in real clinical practice: a randomized, open-label, parallel-group, multi-center study: the DISCOVERY-Beta study
- PMID: 19337553
- PMCID: PMC2663459
- DOI: 10.2147/vhrm.s4151
Achieving lipid goals with rosuvastatin compared with simvastatin in high risk patients in real clinical practice: a randomized, open-label, parallel-group, multi-center study: the DISCOVERY-Beta study
Abstract
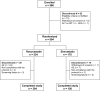
The aim of this multi-center, open-label, randomized, parallel-group trial was to compare the efficacy of rosuvastatin with that of simvastatin in achieving the 1998 European Atherosclerosis Society (EAS) lipid treatment goals. 504 patients (> or =18 years) with primary hypercholesterolemia and a 10-year cardiovascular (CV) risk >20% or history of coronary heart disease (CHD) or other established atherosclerotic disease were randomized in a 2:1 ratio to receive rosuvastatin 10 mg or simvastatin 20 mg once daily for 12 weeks. A significantly higher proportion of patients achieved 1998 EAS low-density lipoprotein cholesterol (LDL-C) goal after 12 weeks of treatment with rosuvastatin 10 mg compared to simvastatin 20 mg (64 vs 51.5%, p < 0.01). Similarly, significantly more patients achieved the 1998 EAS total cholesterol (TC) goal and the 2003 EAS LDL-C and TC goals (p < 0.001) with rosuvastatin 10 mg compared with simvastatin 20 mg. The incidence of adverse events and the proportion of patients who discontinued study treatment were similar between treatment groups. In conclusion, in the DISCOVERY-Beta Study in patients with primary hypercholesterolemia greater proportion of patients in the rosuvastatin 10 mg group achieved the EAS LDL-C treatment goal compared with the simvastatin 20 mg group. Drug tolerability was similar across both treatment groups.
Keywords: hypercholesterolemia; low-density lipoprotein cholesterol; rosuvastatin; simvastatin; statins; tolerability.
Figures
References
-
- Binbrek AS, Elis A, Al-Zaibag M, et al. for the DISCOVERY Alpha Study Group Rosuvastatin versus atorvastatin in achieving lipid goals in patients at high risk for cardiovascular disease in clinical practice: a randomized, open-label, parallel-group, multicenter study (DISCOVERY Alpha Study) Curr Ther Res. 2006;67:21–43. - PMC - PubMed
-
- Bots AFE, Kastelein JJP. Achieving lipid goals in real life: the Dutch DISCOVERY Study. Int J Clin Pract. 2005;59:1387–94. - PubMed
-
- Brown WV, Bays HE, Hassmann DR, et al. Efficacy and safety of rosuvastatin compared with pravastatin and simvastatin in patients with hypercholesterolemia: a randomized, double-blind, 52-week trial. Am Heart J. 2002;144:1036–43. - PubMed
-
- Coresh J, Kwiterovich PO., Jr Small, dense low-density lipoprotein particles and coronary heart disease risk: a clear association with uncertain implications. JAMA. 1996;276:914–5. - PubMed
-
- Davidson M, Ma P, Stein EA, et al. Comparison of effects on low-density lipoprotein cholesterol and high-density lipoprotein cholesterol with rosuvastatin versus atorvastatin in patients with type IIa or IIb hypercholesterolemia. Am J Cardiol. 2002;89:268–75. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical