Role of nuclear-encoded subunit Vb in the assembly and stability of cytochrome c oxidase complex: implications in mitochondrial dysfunction and ROS production
- PMID: 19338496
- PMCID: PMC2735414
- DOI: 10.1042/BJ20090214
Role of nuclear-encoded subunit Vb in the assembly and stability of cytochrome c oxidase complex: implications in mitochondrial dysfunction and ROS production
Abstract
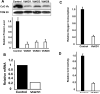
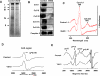
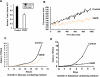
CcO (cytochrome c oxidase) is a multisubunit bigenomic protein complex which catalyses the last step of the mitochondrial electron transport chain. The nuclear-encoded subunits are thought to have roles either in regulation or in the structural stability of the enzyme. Subunit Vb is a peripheral nuclear-encoded subunit of mammalian CcO that is dramatically reduced under hypoxia. Although it has been shown to contain different ligand-binding sites and undergo modifications, its precise function is not known. In the present study we generated a cell line from RAW 264.7 murine macrophages that has a more than 80% reduced level of Vb. Functional analysis of these cells showed a loss of CcO activity, membrane potential and less ability to generate ATP. Resolution of complexes on blue native gel and two-dimensional electrophoretic analysis showed an accumulation of subcomplexes of CcO and also reduced association with supercomplexes of the electron transfer chain. Furthermore, the mitochondria from CcO Vb knock-down cells generated increased ROS (reactive oxygen species), and the cells were unable to grow on galactose-containing medium. Pulse-chase experiments suggest the role of the CcO Vb subunit in the assembly of the complex. We show for the first time the role of a peripheral, non-transmembrane subunit in the formation as well as function of the terminal CcO complex.
Figures




References
-
- Brunori M, Antonini G, Malatesta F, Sarti P, Wilson MT. Cytochrome-c oxidase. Subunit structure and proton pumping. Eur.J.Biochem. 1987;169:1–8. - PubMed
-
- Keightley JA, Hoffbuhr KC, Burton MD, Salas VM, Johnston WS, Penn AM, Buist NR, Kennaway NG. A microdeletion in cytochrome c oxidase (COX) subunit III associated with COX deficiency and recurrent myoglobinuria. Nat.Genet. 1996;12:410–416. - PubMed
-
- Wikstrom MK. Proton pump coupled to cytochrome c oxidase in mitochondria. Nature. 1977;266:271–273. - PubMed
-
- Capaldi RA. Structure and function of cytochrome c oxidase. Annu.Rev.Biochem. 1990;59:569–596. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

