Loss of cone photoreceptors caused by chromophore depletion is partially prevented by the artificial chromophore pro-drug, 9-cis-retinyl acetate
- PMID: 19339306
- PMCID: PMC2685761
- DOI: 10.1093/hmg/ddp163
Loss of cone photoreceptors caused by chromophore depletion is partially prevented by the artificial chromophore pro-drug, 9-cis-retinyl acetate
Abstract
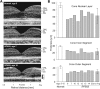
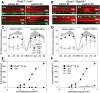
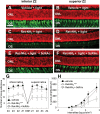
Inactivating mutations in the retinoid isomerase (RPE65) or lecithin:retinol acyltransferase (LRAT) genes cause Leber congenital amaurosis (LCA), a severe visual impairment in humans. Both enzymes participate in the retinoid (visual) cycle, the enzymatic pathway that continuously generates 11-cis-retinal, the chromophore of visual pigments in rod and cone photoreceptor cells needed for vision. We investigated human RPE65-LCA patients and mice with visual cycle abnormalities to determine the impact of chronic chromophore deprivation on cones. Young patients with RPE65 mutations showed foveal cone loss along with shortened inner and outer segments of remaining cones; cone cell loss also was dramatic in young mice lacking Rpe65 or Lrat gene function. To selectively evaluate cone pathophysiology, we eliminated the rod contribution to electroretinographic (ERG) responses by generating double knockout mice lacking Lrat or Rpe65 together with an inactivated rod-specific G protein transducin gene (Gnat1-/-). Cone ERG responses were absent in Gnat1-/-Lrat-/- mice which also showed progressive degeneration of cones. Cone ERG responses in Gnat1-/-Rpe65-/- mice were markedly reduced and declined over weeks. Treatment of these mice with the artificial chromophore pro-drug, 9-cis-retinyl acetate, partially protected inferior retinal cones as evidenced by improved ERGs and retinal histochemistry. Gnat1-/- mice chronically treated with retinylamine, a selective inhibitor of RPE65, also showed a decline in the number of cones that was ameliorated by 9-cis-retinyl acetate. These results suggest that chronic lack of chromophore leads to progressive loss of cones in mice and humans. Therapy for LCA patients should be geared toward early adequate delivery of chromophore to cone photoreceptors.
Figures


Similar articles
-
Trafficking of membrane-associated proteins to cone photoreceptor outer segments requires the chromophore 11-cis-retinal.J Neurosci. 2008 Apr 9;28(15):4008-14. doi: 10.1523/JNEUROSCI.0317-08.2008. J Neurosci. 2008. PMID: 18400900 Free PMC article.
-
Bax-induced apoptosis in Leber's congenital amaurosis: a dual role in rod and cone degeneration.PLoS One. 2009 Aug 12;4(8):e6616. doi: 10.1371/journal.pone.0006616. PLoS One. 2009. PMID: 19672311 Free PMC article.
-
Pharmacological Amelioration of Cone Survival and Vision in a Mouse Model for Leber Congenital Amaurosis.J Neurosci. 2016 May 25;36(21):5808-19. doi: 10.1523/JNEUROSCI.3857-15.2016. J Neurosci. 2016. PMID: 27225770 Free PMC article.
-
Vitamin A derivatives as treatment options for retinal degenerative diseases.Nutrients. 2013 Jul 12;5(7):2646-66. doi: 10.3390/nu5072646. Nutrients. 2013. PMID: 23857173 Free PMC article. Review.
-
Lecithin:Retinol Acyltransferase: A Key Enzyme Involved in the Retinoid (visual) Cycle.Biochemistry. 2016 Jun 7;55(22):3082-91. doi: 10.1021/acs.biochem.6b00319. Epub 2016 May 23. Biochemistry. 2016. PMID: 27183166 Free PMC article. Review.
Cited by
-
M-opsin protein degradation is inhibited by MG-132 in Rpe65⁻/⁻ retinal explant culture.Mol Vis. 2012;18:1516-25. Epub 2012 Jun 13. Mol Vis. 2012. PMID: 22736942 Free PMC article.
-
Lipofuscin causes atypical necroptosis through lysosomal membrane permeabilization.Proc Natl Acad Sci U S A. 2021 Nov 23;118(47):e2100122118. doi: 10.1073/pnas.2100122118. Proc Natl Acad Sci U S A. 2021. PMID: 34782457 Free PMC article.
-
Retinoid isomerase inhibitors impair but do not block mammalian cone photoreceptor function.J Gen Physiol. 2018 Apr 2;150(4):571-590. doi: 10.1085/jgp.201711815. Epub 2018 Mar 2. J Gen Physiol. 2018. PMID: 29500274 Free PMC article.
-
Proceedings of the First International Optogenetic Therapies for Vision Symposium.Transl Vis Sci Technol. 2013 Nov;2(7):4. doi: 10.1167/tvst.2.7.4. Epub 2013 Nov 21. Transl Vis Sci Technol. 2013. PMID: 24349882 Free PMC article.
-
TULP1 mutations causing early-onset retinal degeneration: preserved but insensitive macular cones.Invest Ophthalmol Vis Sci. 2014 Jul 29;55(8):5354-64. doi: 10.1167/iovs.14-14570. Invest Ophthalmol Vis Sci. 2014. PMID: 25074776 Free PMC article.
References
-
- Stone E.M. Leber congenital amaurosis—a model for efficient genetic testing of heterogeneous disorders: LXIV Edward Jackson Memorial Lecture. Am. J. Ophthalmol. 2007;144:791–811. - PubMed
-
- den Hollander A.I., Roepman R., Koenekoop R.K., Cremers F.P. Leber congenital amaurosis: genes, proteins and disease mechanisms. Prog. Retin. Eye Res. 2008;27:391–419. - PubMed
-
- Redmond T.M., Yu S., Lee E., Bok D., Hamasaki D., Chen N., Goletz P., Ma J.X., Crouch R.K., Pfeifer K. Rpe65 is necessary for production of 11-cis-vitamin A in the retinal visual cycle. Nat. Genet. 1998;20:344–351. - PubMed
-
- Aguirre G.D., Baldwin V., Pearce-Kelling S., Narfstrom K., Ray K., Acland G.M. Congenital stationary night blindness in the dog: common mutation in the RPE65 gene indicates founder effect. Mol. Vis. 1998;4:23. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

