Late Na+ current produced by human cardiac Na+ channel isoform Nav1.5 is modulated by its beta1 subunit
- PMID: 19340536
- PMCID: PMC2744134
- DOI: 10.1007/s12576-009-0029-7
Late Na+ current produced by human cardiac Na+ channel isoform Nav1.5 is modulated by its beta1 subunit
Abstract
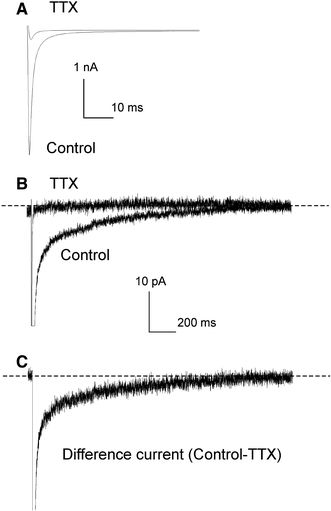
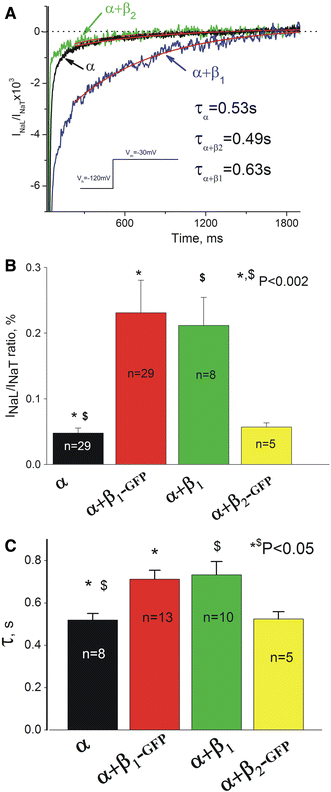
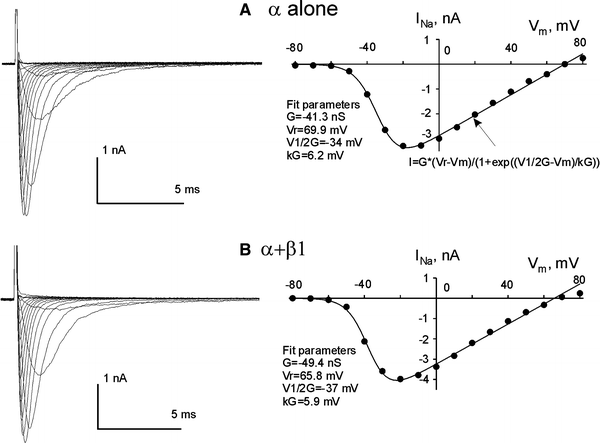
Experimental data accumulated over the past decade show the emerging importance of the late sodium current (I(NaL)) for the function of both normal and, especially, failing myocardium, in which I(NaL) is reportedly increased. While recent molecular studies identified the cardiac Na(+) channel (NaCh) alpha subunit isoform (Na(v)1.5) as a major contributor to I (NaL), the molecular mechanisms underlying alterations of I(NaL) in heart failure (HF) are still unknown. Here we tested the hypothesis that I(NaL) is modulated by the NaCh auxiliary beta subunits. tsA201 cells were transfected simultaneously with human Na(v)1.5 (former hH1a) and cardiac beta(1) or beta(2) subunits, and whole-cell patch-clamp experiments were performed. We found that I(NaL) decay kinetics were significantly slower in cells expressing alpha + beta(1) (time constant tau = 0.73 +/- 0.16 s, n = 14, mean +/- SEM, P < 0.05) but remained unchanged in cells expressing alpha + beta(2) (tau = 0.52 +/- 0.09 s, n = 5), compared with cells expressing Na(v)1.5 alone (tau = 0.54 +/- 0.09 s, n = 20). Also, beta(1), but not beta(2), dramatically increased I(NaL) relative to the maximum peak current, I(NaT) (2.3 +/- 0.48%, n = 14 vs. 0.48 +/- 0.07%, n = 6, P < 0.05, respectively) and produced a rightward shift of the steady-state availability curve. We conclude that the auxiliary beta(1) subunit modulates I(NaL), produced by the human cardiac Na(+) channel Na(v)1.5 by slowing its decay and increasing I(NaL) amplitude relative to I(NaT). Because expression of Na(v)1.5 reportedly decreases but beta(1) remains unchanged in chronic HF, the relatively higher expression of beta(1) may contribute to the known I(NaL) increase in HF via the modulation mechanism found in this study.
Figures

Similar articles
-
Post-transcriptional alterations in the expression of cardiac Na+ channel subunits in chronic heart failure.J Mol Cell Cardiol. 2004 Jul;37(1):91-100. doi: 10.1016/j.yjmcc.2004.04.003. J Mol Cell Cardiol. 2004. PMID: 15242739 Free PMC article.
-
Molecular identity of the late sodium current in adult dog cardiomyocytes identified by Nav1.5 antisense inhibition.Am J Physiol Heart Circ Physiol. 2008 Aug;295(2):H667-76. doi: 10.1152/ajpheart.00111.2008. Epub 2008 Jun 13. Am J Physiol Heart Circ Physiol. 2008. PMID: 18552167 Free PMC article.
-
Post-transcriptional silencing of SCN1B and SCN2B genes modulates late sodium current in cardiac myocytes from normal dogs and dogs with chronic heart failure.Am J Physiol Heart Circ Physiol. 2011 Oct;301(4):H1596-605. doi: 10.1152/ajpheart.00948.2009. Epub 2011 Jun 24. Am J Physiol Heart Circ Physiol. 2011. PMID: 21705762 Free PMC article.
-
Late sodium current is a new therapeutic target to improve contractility and rhythm in failing heart.Cardiovasc Hematol Agents Med Chem. 2008 Oct;6(4):348-59. doi: 10.2174/187152508785909447. Cardiovasc Hematol Agents Med Chem. 2008. PMID: 18855648 Free PMC article. Review.
-
Late sodium current in failing heart: friend or foe?Prog Biophys Mol Biol. 2008 Jan-Apr;96(1-3):421-51. doi: 10.1016/j.pbiomolbio.2007.07.010. Epub 2007 Aug 10. Prog Biophys Mol Biol. 2008. PMID: 17854868 Free PMC article. Review.
Cited by
-
Cardioprotection by SGLT2 Inhibitors-Does It All Come Down to Na+?Int J Mol Sci. 2021 Jul 26;22(15):7976. doi: 10.3390/ijms22157976. Int J Mol Sci. 2021. PMID: 34360742 Free PMC article. Review.
-
Ca2+/calmodulin-dependent protein kinase II (CaMKII) regulates cardiac sodium channel NaV1.5 gating by multiple phosphorylation sites.J Biol Chem. 2012 Jun 8;287(24):19856-69. doi: 10.1074/jbc.M111.322537. Epub 2012 Apr 18. J Biol Chem. 2012. PMID: 22514276 Free PMC article.
-
CaMKII-dependent regulation of cardiac Na(+) homeostasis.Front Pharmacol. 2014 Mar 10;5:41. doi: 10.3389/fphar.2014.00041. eCollection 2014. Front Pharmacol. 2014. PMID: 24653702 Free PMC article. Review.
-
Mechanisms of noncovalent β subunit regulation of NaV channel gating.J Gen Physiol. 2017 Aug 7;149(8):813-831. doi: 10.1085/jgp.201711802. J Gen Physiol. 2017. PMID: 28720590 Free PMC article.
-
Fibroblast growth factor homologous factors serve as a molecular rheostat in tuning arrhythmogenic cardiac late sodium current.Nat Cardiovasc Res. 2022 May;1(5):1-13. doi: 10.1038/s44161-022-00060-6. Epub 2022 May 16. Nat Cardiovasc Res. 2022. PMID: 35662881 Free PMC article.
References
-
- Maltsev VA, Sabbah HN, Tanimura M, Lesch M, Goldstein S, Undrovinas AI. Relationship between action potential, contraction-relaxation pattern, and intracellular Ca2+ transient in cardiomyocytes of dogs with chronic heart failure. Cell Mol Life Sci. 1998;54:597–605. doi: 10.1007/s000180050187. - DOI - PMC - PubMed
-
- Undrovinas AI, Belardinelli L, Undrovinas NA, Sabbah HN. Ranolazine improves abnormal repolarization and contraction in left ventricular myocytes of dogs with heart failure by inhibiting late sodium current. J Cardiovasc Electrophysiol. 2006;17:S169–S177. doi: 10.1111/j.1540-8167.2006.00401.x. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

